Educational aims and objectives
This self-instructional course for dentists aims to show information and techniques regarding maintenance of implants for both in-office and at-home processes.
Expected outcomes
Implant Practice US subscribers can answer the CE questions by taking the quiz to earn 2 hours of CE from reading this article.
Correctly answering the questions will demonstrate the reader can:
- Identify different instrument materials that reduce damage to dental implants.
- Realize the positive role disclosing solutions can make on biofilm education.
- Recognize the importance of implant education for patients with implants.
- Identify some characteristics of air-polishing technology.
- Identify some at-home care products that have benefits for dental implants.
In part 2 of their CE, Dr. Gregori M. Kurtzman and Debbie Zafiropoulos discuss dental hygiene in the office and at home that can help maintain implant longevity.
Dr. Gregori M. Kurtzman, along with Debbie Zafiropoulos, discusses in-office and at-home dental implant care
Implant maintenance requires both professional services (routine recall appointments) as well as good home care by the patient to minimize issues related to biofilm accumulation and the resulting peri-mucositis that can result. Cleaning of implants and their prosthetics need different approaches than natural teeth and their prosthetics.
Professional cleaning of instrumentation: an overview
Instruments made of metal such as stainless steel should be limited to use on natural teeth and not to probe or scale dental implants. The rationale for this well-documented conclusion is that the hardness of this metal can cause scratches, contamination, or a galvanic reaction at the implant-abutment interface.1,2
Ideally, hand periodontal scalers for cleaning dental implants can be made of plastic, Teflon™, gold-plated, or wood (Figures 1 and 2).3,4 When using gold-plated curettes, the manufacturer recommends not sharpening these hygiene instruments, as the gold surface could be chipped exposing the hard metal underneath this coating. Stainless steel scaling instruments may abrade the implant surface, stripping off any surface treatment such as hydroxyapatite (HA) as the instrument’s hardness is greater than the titanium alloy the implant is fabricated from (Figure 3).
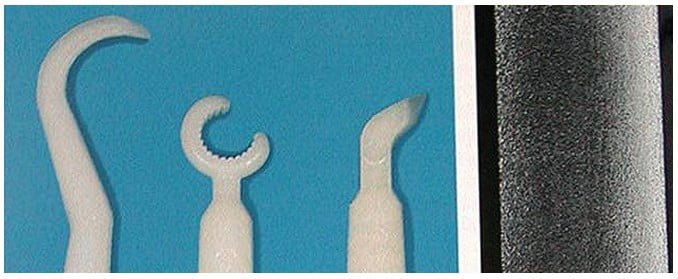
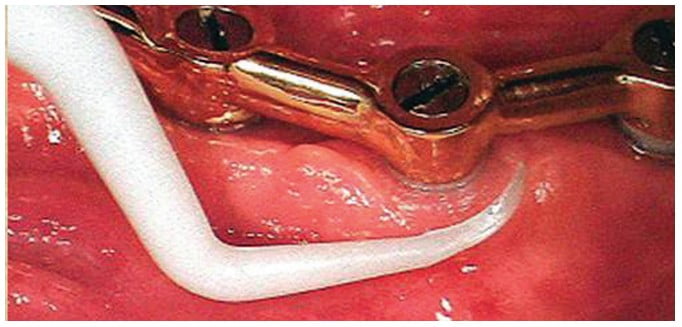
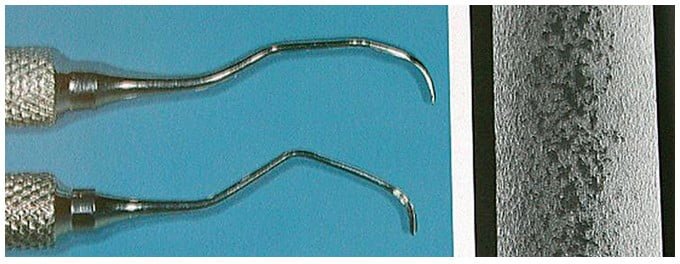
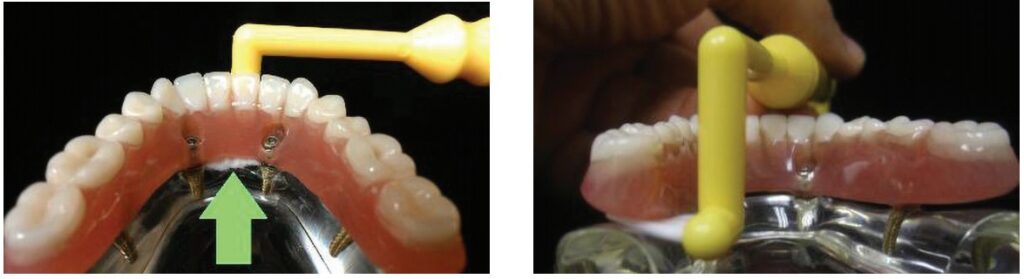
Other cleaning armamentarium contraindicated for use with dental implants are air powder abrasive units, flour or pumice for polishing, and sonic and ultrasonic scaling units.5,6 Ultrasonic, piezo, or sonic scaler tips may mar the implant’s surface leading to microroughness and plaque accumulation. The stainless steel tip may also lead to gouging of the implant’s polished collar (Figure 4). However, some clinicians advocate using a sonic instrument with a plastic sleeve over the tip for scaling dental implants. Air powder polishing units may also damage the implant surface and should be avoided during hygiene appointments (Figure 5). Even the use of baking soda powder in these units may strip off any surface coating on the implant. Additionally, the air pressure may detach the soft tissue connection with the coronal of the implant possibly leading to emphysema. Titanium or titanium alloy surfaces of dental implants can be polished using a rubber cup along with a nonabrasive polishing paste or a gauze strip with tin oxide.


Improving implant recall prophy maintenance with disclosing solution
Why disclose?
Dental healthcare professionals often equate the use of disclosing solution as undesirable as it can be time-consuming, difficult to manage, limited to pediatric patients, or merely utilized in an academic clinical setting. However, the application of disclosing solution as part of preventive maintenance workflow is an essential step in identifying biofilm and is beneficial for both the patient and clinician.
While disclosing solutions have been tucked away in our operatory cabinets, products have improved over the years, which has simplified the application process and aligns vital patient education with the oral-systemic link and, subsequently, improving patient compliance. Medical science has well documented the impact oral biofilm has on long-term dental implant health and stability, which indicates that facilitating biofilm management strategies in implant maintenance protocols is a critical component of the dental hygiene process of care.7-9
Oral and systemic health
Medical scientific evidence continues to correlate the influence of pathogenic oral biofilm on noncommunicable systemic diseases. Embracing the understanding that preventive dental care and patient education have on reducing the impact of active disease, managing disease, and preventing disease through dental biofilm disruption is necessary for periodontal and peri-implant health. The periodontal pathogen, P gingivalis, has been linked to many noncommunicable diseases such as cardiovascular disease, diabetes, Alzheimer’s, rheumatoid arthritis, kidney disease, erectile dysfunction, chronic inflammation, and increased risk for some cancers.10-16 Since 42% of Americans have periodontitis, dental professionals need to recognize this scientific evidence to implement protocols in prevention that meet the needs of this type of patient and adopt a comprehensive methodology to preventive dental care.17
Dental implant patient education
Dental implant patients require maintenance that extends beyond traditional preventive protocols. Dental professionals have a responsibility to recognize the importance of technologies and clinical workflows tailored for implant maintenance to aide in the disruption of oral biofilm and its associated inflammation for implant health and success. As patient compliance ranks as the most significant barrier to improving patients’ oral health, disclosing solution lends the ability for dental professionals to educate patients of the areas where self-care efforts require improvement. The use of disclosing agents will allow dental professionals the opportunity to inform patients of the consequences of biofilm dysbiosis to dental and systemic health, personalize home care to the specific needs of the patient, and demonstrate the efficacy of home care recommendations through a hands-on approach to gain an understanding of the disease process induced by patient noncompliance.18,19
Disclosing application
The application of disclosing solution has improved for ease of use and the ability to identify different maturity levels of bacteria. Contemporary disclosing solutions come in gel form, pre-loaded applicators, and presoaked sponges (Figures 6-9). The gel solution form may be applied with a cotton applicator, applicator brush, or toothbrush (Figures 10 and 11).
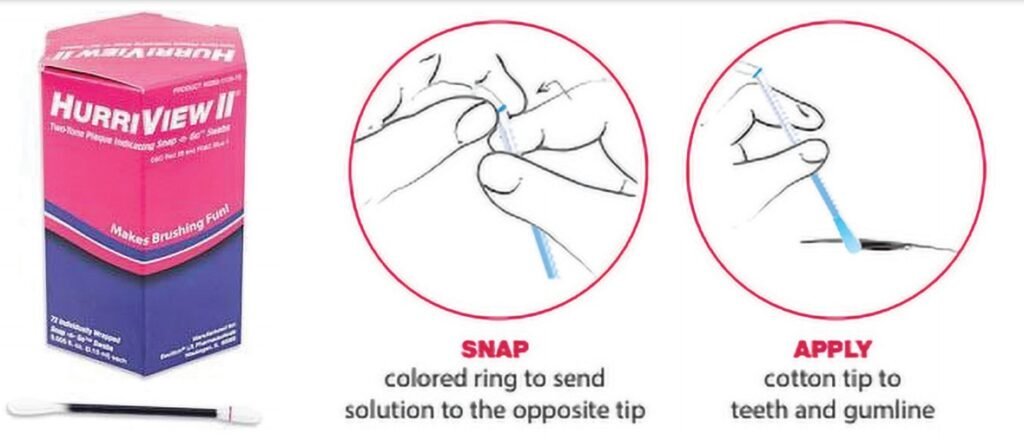
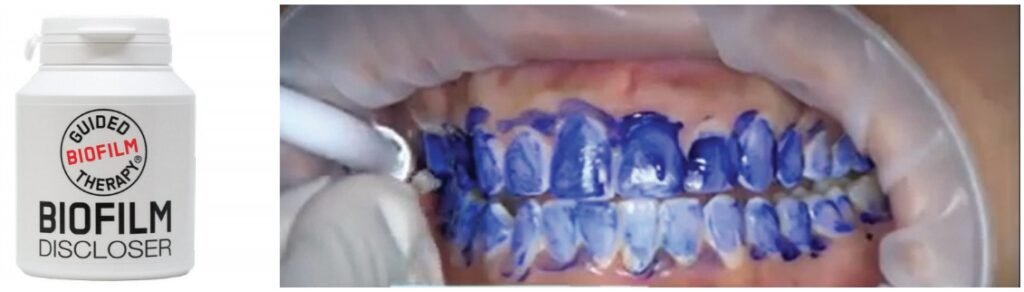
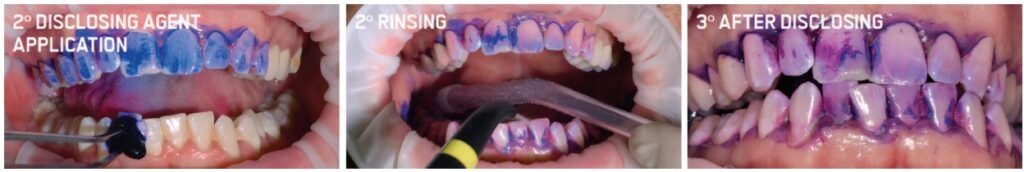
Once the disclosing solution is applied, have the patient rinse, and then review the results. Educating patients to the different colors that result related to the age and virulence of bacteria enables an understanding of periodontal and peri-implant disease etiology.
Performing home care instructions before debridement helps patients visualize the biofilm removal process and efficacy. After the demonstration, have the patient perform the given instructions and coach as needed. Once education and home care instructions are complete, then debridement procedures can begin. This small shift in the clinical workflow will ensure that critical patient education is routinely included in maintenance appointments, thus leading to improved patient outcomes and increased implant health and success.
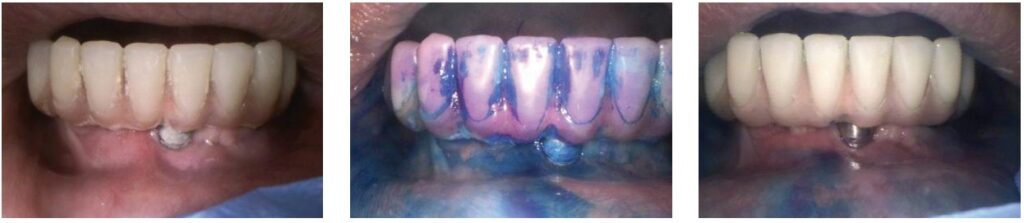
Furthermore, when applying the Guided Biofilm Therapy (GBT) eight-step protocol to implant maintenance, debridement outcomes improve as the clinician has a roadmap for biofilm and calculus removal via disclosing solution (Figures 12-14). The GBT protocol allows for more thorough care, less labor-intensive work for the clinician, and a minimally invasive approach to maintaining dental implant surface integrity.20
Improving oral biofilm in-office removal via air polishing
Air polishing with glycine powder may be considered as a better method to remove plaque from dental implant because glycine is less aggressive than sodium bicarbonate powder. Moreover, the use of glycine powder seems to have an active role on the inhibition of bacterial recolonization of implants in a short test period (24 hours).21 Other studies indicate that glycine powder assists in the reduction of bleeding upon probing after nonsurgical therapy. This is a key point that would entice the clinician/patient communication of utilizing noninvasive therapy to reduce bleeding.22
In the past few years, indications for the use of the air-polishing technology have been expanded from supragingival use (airflow) to subgingival air polishing (PERIOFLOW) by the development of new low-abrasive glycine-based powders and devices with a subgingival nozzle. Several studies on the subgingival use of air polishing have been completed. In June of 2012, during the Europerio 7 Congress in Vienna, a consensus conference on mechanical biofilm management took place aiming to review the current evidence from the literature on the clinical relevance of the subgingival use of air polishing and to make practical recommendations for the clinician.23 Their consensus concluded that:
- Oral disease is caused by biofilm.
- Mechanical bacterial biofilm management is essential to longevity.
- Air-polishing devices are effective in removing supra and subgingival biofilm and stain.
- Air-polishing devices shorten the treatment time.
- Air polishing diminishes root sensitivity compared to stainless steel
In order for the restorative clinician (dentist) and the maintenance therapist (hygienist) to successfully collaborate in the long-term health and stability of the prothesis, it is imperative that they first identify any risk factors that inhibit a successful procedure through all steps. Understanding the integrity of the implant surface is key to long-term maintenance of the implants intraorally, and optimized in-office therapies can be provided by the dental practitioners (dentists and hygienists) and the patient during at home maintenance protocols. The goal is to minimize iatrogenic damage to the implant surface which can happen with the use of metal hand or ultrasonic instruments and diminish pathogenic biofilm adhesion to the implant surfaces that are supracrestal and the prosthetics. Once this is established, the patient should be enrolled in long-term biofilm therapy to ensure preventive management of the implant and prosthesis. The GBT Compass optimizes the patient/clinician appointment time in a synchronized manner to enhance assessment, patient motivation and home care protocols as well as more effective, noninvasive biofilm therapy with both patient and clinician comfort (Figure 15).
Traditional air polishers use higher pressure as discussed previously, but units have been developed that utilize very low air pressure so the potential issues encountered and reported with the older units are not observed in the newer implant specific air polishers. When it comes to the opportunity to use Airflow (EMS, Nyon, Switzerland), one of the benefits is the use of erythritol powder. In the past, air polishers have been limited to bicarbonate or glycine. While each powder has its limitations, the use of erythritol has overwhelming attributes that contribute to its safe and comfortable application. Erythritol is a polyol (sugar alcohol) that has a smaller particle size of 14µm in comparison to sodium bicarbonate (40µm) or glycine (25µm). Due to the smaller particle size and decreased abrasiveness, the advantages of erythritol in GBT therapy allows the clinician to comfortably deliver supra and subgingival airflow therapy with increased comfort to the patient without damaging the cementum. Usage around implants that lie surpracrestally allow more thorough removal of biofilm on the surfaces without the potential for roughening the exposed titanium and becoming a future biofilm accumulation area (Figure 16). Additionally, airflow is able to reach areas not easily accessible between the inferior surface of the prosthesis and the soft tissue (Figure 17). Erythritol’s hardness factor and smaller particle size also allows the clinician to remove more stain with less powder volume than glycine which cuts down on per treatment costs. Additionally, erythritol has been shown to have an antimicrobial inhibition factor for S gordonii and P gingivalis.24


Based on the most recent marketing reports, dental implants and prosthesis market is expected to reach $13.01 billion USD by 2023, up 6.5% from an estimated $9.50 billion in 2018. Key factors driving the growth of this market include the growing geriatric population and corresponding dental disorders, including the rising incidence of tooth decay and edentulism across the globe, growing dental tourism in emerging markets, and increasing government expenditure on oral healthcare.25
PERIOFLOW®
Peri-implantitis has been defined as an emerging public health issue. According to recent scientific data and depending on the clinical and radiographic criteria that are adopted, it can affect from 20% to 40% of subjects restored with dental implants.
The PERIOFLOW® device (EMS Dental) provides a flexible nozzle and lightweight body providing a minimally invasive treatment of periodontal pockets with a uniform trilateral powder spray (Figure 18). The flexible tip can be inserted atraumatically into the sulcus (Figure 19). Three outlets at the tip provide comfortable decontamination of the tissue, the implant, and the base of the sulcus with erythritol powder delivered with warmed water with the PERIOFLOW handpiece as part of GBT therapy. It’s ideal for supportive periodontal therapy (SPT), primary and secondary prevention, as well as implant maintenance and nonsurgical treatment of peri-implantitis and periodontitis.
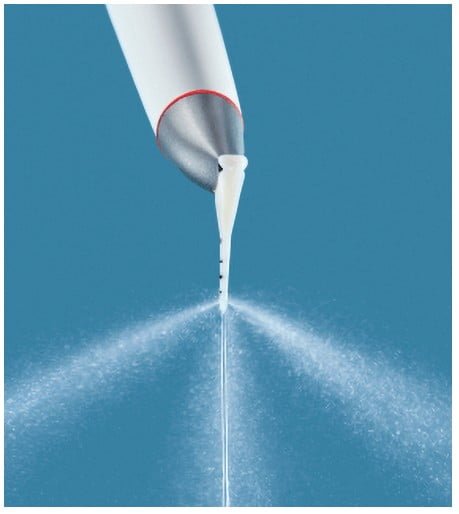
Home care
Dental implants require special home care. Clinicians should discourage the use of normal OTC toothpastes, because their ingredients can damage the implant surfaces. Fluoride, pH-value, abrasives, and bad oral hygiene can all affect the prosthesis by affecting the implant and prosthesis surfaces, increasing their biofilm adhesion, and thus contributing to peri-mucositis that may progress to peri-implantitis. Ideal medicaments should be recommended based on the patient’s risk factors, commitment to optimized home care protocols and other comorbidities. Ideally the product should not be abrasive, maintain a neutral pH-value, and minimal amount of fluoride to prevent damaging the implant.
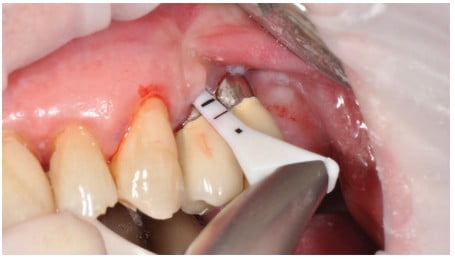
Patients should be taught the modified bass technique of brushing using a medium-sized head, soft-bristled toothbrush. The use of intradental brushes should be used by implant patients after being shown their proper use. The plastic-coated wire brush is the only type to be used with dental implants to clean and not scratch the implant surface (Figure 20).

A challenge with home care with regard to partially or fully edentulous implant prosthetics is difficulty for the patient to access the surfaces that can harbor biofilm. Traditional toothbrushes do not permit access to these areas specifically between the fixed prosthesis and gingival tissue and on the lingual aspects. A modified specialty toothbrush designed to access areas that are difficult to clean with a traditional brush, the ImplantClean™ Brush (www.implantclean.com) permits easier home care for the patient in those areas in and around the prosthetics (Figure 21). The angulation of the bristles allows better penetration into the sulcus around single-unit restored implants (as well as around non-splinted natural teeth) to remove subgingival biofilm (Figure 22-24). Lingual aspects of the posterior teeth, especially those with wider emergence profiles compared to the implant’s diameter or under cantilever pontics are easier to access (Figure 25). The increased frequency of hybrid prosthetics presents challenges to the patient regarding home care. These prostheses often have cantilever posterior pontics and are adapted to flat ridges creating potential food traps and biofilm accumulation areas. The angulation of the bristles in relation to the handle on the ImplantClean Brush allow better cleaning between the inferior aspect of the hybrid and the soft tissue, with the bristles being able to pass through fully to the lingual aspect of the arch (Figure 26). The inferior aspect of the posterior cantilevers can be accessed either from the lingual (Figure 27) or buccal giving the patient a choice as to which is easier when cleaning that aspect of the bridge.
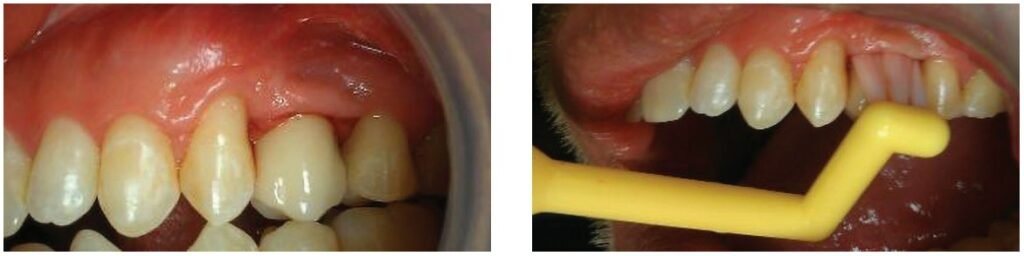
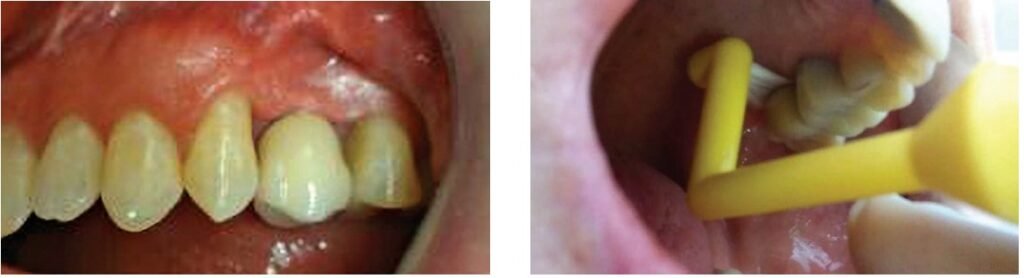
It is difficult to get a patient to brush longer, but if we can improve efficiency in the time they actually brush, we can improve the quality of their home care. Electric toothbrushes have been advocated as a daily mode of tooth cleansing with natural teeth and they improve home care with implants too. These devices may be a rotary, circular, or sonic type. The key to the effectiveness of these home-care instruments is proper instruction on their use and then diligent daily use by the implant patient. Specialized manual toothbrushes have been developed that are designed to access those areas that a traditional manual or electric toothbrush may not be able to access. This becomes particularly difficult for the patient with a hybrid prosthesis.
As with natural dentition, adjunctive cleaning aids such as flossing are still valuable. As with dentated patients, an implant patient’s home care requirements should be individually tailored according to each patient’s needs. Individual needs are based on the location and angulation of the dental implants, the position and length of transmucosal abutments, the type of prosthesis, and the dexterity of each patient.
The other popularized type of cleansing device is the use of oral irrigators with or without the addition of antimicrobial solutions. Also, oral rinses with antimicrobial properties such as Listerine or chlorhexidine have been widely advocated throughout the literature.31-33
Conclusion
During the early years of dental implantology, the emphasis for long-term success of osseointegrated implants was the surgical phase of dental implantology. In the years that followed, the emphasis for success had switched from a purely surgical influence to focusing more on the proper fixture placement, which would be dictated by the prosthetic and esthetic needs of each particular case.
The dental professional has recognized professional implant maintenance and diligent patient home care as two critical factors for the long-term success of dental implants. The microbiota and clinical presentation of peri-implantitis is the same as periodontitis around a natural tooth. Elimination of oral biofilm is key to preventing peri-mucositis that can progress to peri-implantitis with potential loss of the implants. This falls on both the dental professional at recall appointments and the patient via good home care.
Read part 1 of “Oral hygiene and dental implant maintenance” at https://implantpracticeus.com/ce-articles/oral-hygiene-and-dental-implant-maintenance-part-1/
References
- Speelman JA, Collaert B, Klinge B. Evaluation of different methods to clean titanium abutments. A scanning electron microscopic study. Clin Oral Implants Res. 1992;3(3):120-127.
- Louropoulou A, Slot DE, Van der Weijden F. Influence of mechanical instruments on the biocompatibility of titanium dental implants surfaces: a systematic review. Clin Oral Implants Res. 2015;26(7):841-850.
- Louropoulou A, Slot DE, Van der Weijden FA. Titanium surface alterations following the use of different mechanical instruments: a systematic review. Clin Oral Implants Res. 2012;23(6):643-658.
- Gantes BG, Nilveus R. The effects of different hygiene instruments on titanium surfaces: SEM observations. Int J Periodontics Restorative Dent. 1991;11(3):225-239.
- Rapley JW, Swan RH, Hallmon WW, et al. The oral hygiene instruments and materials on titanium implant abutments. Int J Oral Maxillofac Implants. 1990;5(1):47-52.
- Unursaikhan O, Lee JS, Cha JK, et al. Comparative evaluation of roughness of titanium surfaces treated by different hygiene instruments. J Periodontal Implant Sci. 2012;42(3):88-94.
- Heitz-Mayfield, LJA, Salvi, GE. Peri-implant mucositis. J Clin Periodontol.2018; 45(suppl 20):S237-S245.
- Schwarz, F, Derks, J, Monje, A, Wang, HL. Peri-implantitis.J Clin Periodontol. 2018; 45(suppl 20):S246-S266.
- Lasserre JF, Brecx MC, Toma S. Oral Microbes, Biofilms and Their Role in Periodontal and Peri-Implant Diseases. Materials (Basel). 2018;11(10):1802.
- Aguilera EM, Suvan J, Buti J, et al. Periodontitis is associated with hypertension. A systematic review and meta-analysis. Cardiovasc Res. 2020;116(1):28-39.
- Zeng XT, Xia LY, Zhang YG, et al. Periodontal Disease and Incident Lung Cancer Risk: A Meta-Analysis of Cohort Studies. J Periodontol. 2016;87(10)1158-1164.
- Jacob JA. Study Links Periodontal Disease Bacteria to Pancreatic Cancer Risk. JAMA 2016;315:2653-2654.
- Oğuz F, Eltas A, Beytur A, et al. Is there a relationship between chronic periodontitis and erectile dysfunction? J Sex Med. 2013;10(3):838-843.
- Wahid A, Chaudhry S, Ehsan A, et al. Bidirectional Relationship between Chronic Kidney Disease and Periodontal Disease. Park J Med Science. 2013;29 (1):211-215.
- Rodríguez-Lozano B, González-Febles J, Garnier-Rodríguez JL, et al. Association between severity of periodontitis and clinical activity in rheumatoid arthritis patients: a case-control study. Arthritis Research & Therapy. 2019;21(27).
- Singhrao S. K., Olsen I. Assessing the role of Porphyromonas gingivalis in periodontitis to determine a causative relationship with Alzheimer’s disease. J Oral Microbiol. 2019;11(1):1563410.
- Eke PI, Thornton-Evans GO, Wei L, et al. Periodontitis in US Adults: National Health and Nutrition Examination Survey 2009-2014. J Am Dent Assoc. 2018;149(7):576-588.
- Huang J, Yao Y, Jiang J, Li C. Effects of motivational methods on oral hygiene of orthodontic patients: A systematic review and meta-analysis. Medicine (Baltimore). 2018;97(47):e13182.
- Peng Y, Wu R, Qu W, et al. Effect of visual method vs plaque disclosure in enhancing oral hygiene in adolescents and young adults: a single-blind randomized controlled trial. Am J Orthod Dentofacial Orthop.2014;145(3):280-286
- Louropoulou A, Slot DE, Van der Weijden F. The effects of mechanical instruments on contaminated titanium dental implant surfaces: a systematic review. Clin Oral Implants Res. 2014;25(10):1149-1160.
- Cochis A, Fini M, Carrassi A, et al. Effect of air polishing with glycine powder on titanium abutment surfaces. Clin Oral Implants Res. 2013;24(8):904-909.
- Schwarz F, Becker K, Renvert S. Efficacy of air polishing for the non-surgical treatment of peri-implant diseases: a systematic review. J Clin Periodontal. 2015;42(10):951-959.
- Sculean A, Bastendorf KD, Becker C, et al. A paradigm shift in mechanical biofilm management? Subgingival air polishing: a new way to improve mechanical biofilm management in the dental practice. Quintessence Int. 2013;44(7):475-477.
- Hashino E, Kuboniwa M, Alghamdi S, Yaet al. Erythritol alters microstructure and metabolomic profiles of biofilm composed of Streptococcus gordonii and Porphyromonas gingivalis. Mol Oral Microbiol. 2013;28(6):435-451.
- MarketsandMarkets™ report: Dental Implants and Prosthesis Market by Type (Dental Implants, Bridge, Crown, Abutment, Dentures, Veneers, Inlay & Onlays), Material (Titanium, Zirconium, Metal, Ceramic, Porcelain Fused to Metal), Type of Facility, and Region. Global Forecast to 2023.
Stay Relevant With Implant Practice US
Join our email list for CE courses and webinars, articles and mores

 Gregori M. Kurtzman, DDS, MAGD, FPFA, FACD, FADI, DICOI, DADIA, is in private general practice in Silver Spring, Maryland. He is a former Assistant Clinical Professor at University of Maryland in the department of Restorative Dentistry and Endodontics and a former AAID Implant Maxi-Course assistant program director at Howard University College of Dentistry. He has lectured internationally on the topics of restorative dentistry, endodontics, implant surgery and prosthetics, removable and fixed prosthetics, and periodontics. Dr. Kurtzman has over 750 published articles globally. He has earned Fellowship in the AGD, ACD, ICOI, Pierre Fauchard, ADI, Mastership in the AGD and ICOI, and Diplomate status in the ICOI and American Dental Implant Association (ADIA). A consultant and evaluator for multiple dental companies, Dr. Kurtzman has been honored to be included in the “Top Leaders in Continuing Education” by Dentistry Today annually since 2006. He can be reached at
Gregori M. Kurtzman, DDS, MAGD, FPFA, FACD, FADI, DICOI, DADIA, is in private general practice in Silver Spring, Maryland. He is a former Assistant Clinical Professor at University of Maryland in the department of Restorative Dentistry and Endodontics and a former AAID Implant Maxi-Course assistant program director at Howard University College of Dentistry. He has lectured internationally on the topics of restorative dentistry, endodontics, implant surgery and prosthetics, removable and fixed prosthetics, and periodontics. Dr. Kurtzman has over 750 published articles globally. He has earned Fellowship in the AGD, ACD, ICOI, Pierre Fauchard, ADI, Mastership in the AGD and ICOI, and Diplomate status in the ICOI and American Dental Implant Association (ADIA). A consultant and evaluator for multiple dental companies, Dr. Kurtzman has been honored to be included in the “Top Leaders in Continuing Education” by Dentistry Today annually since 2006. He can be reached at  Debbie Zafiropoulos, EFDA, RDH, is the CEO of the OralED Institute, a Partner in Education for the Wellness Dentistry Network, an instructor with MoradoASC, and certified GBT Trainer for EMS-NA. Zafiropoulos works with top corporate companies in health, creating and delivering live and online educational programs to a worldwide audience. As a sought-after key opinion leader and author, she is determined to deliver programs of forward motion in prevention, science, and technology. In 2016, Zafiropoulos was a recipient of the SUNSTAR Award of Distinction. In 2017, she was recognized as one of the Top 25 Women in Dentistry for her advances in research and prevention of HPV-related oral cancer.
Debbie Zafiropoulos, EFDA, RDH, is the CEO of the OralED Institute, a Partner in Education for the Wellness Dentistry Network, an instructor with MoradoASC, and certified GBT Trainer for EMS-NA. Zafiropoulos works with top corporate companies in health, creating and delivering live and online educational programs to a worldwide audience. As a sought-after key opinion leader and author, she is determined to deliver programs of forward motion in prevention, science, and technology. In 2016, Zafiropoulos was a recipient of the SUNSTAR Award of Distinction. In 2017, she was recognized as one of the Top 25 Women in Dentistry for her advances in research and prevention of HPV-related oral cancer.

