Drs. Jack Krauser and Peter Vitruk explore soft tissue recession prevention with the CO2 laser
 Introduction
Introduction
In 1986, Albrektsson, et al.,1 proposed criteria for evaluating implant success based on clinical and radiographic evidence of osseointegration: the healing of bone around implants to produce direct anchorage of the implant that is then maintained during functional loading without the growth of fibrous tissue at the bone-implant interface.2 The extensive body of peer-reviewed literature published in the field of implantology since then offers a number of additional criteria to define implant success. These criteria include the absence of peri-implantitis, lack of pain and implant mobility, radiographic evidence of minimal crestal bone loss, clinical function, esthetic outcome, and patient satisfaction.1,3-5
 Despite having predictable outcome and long-term success rate, implants sometimes fail — i.e., require removal or have already been lost.5 Implant failures may be classified as early, when the implant body fails to get osseointegrated, or late, when the implant body is unable to sustain the osseo-
Despite having predictable outcome and long-term success rate, implants sometimes fail — i.e., require removal or have already been lost.5 Implant failures may be classified as early, when the implant body fails to get osseointegrated, or late, when the implant body is unable to sustain the osseo-
integration.6 A number of clinical studies have identified various risk factors that may cause or contribute to implant failure.3,5,7 Among the factors associated with implant failures are bone quality and quantity, history of perio-dontal disease, edentulism, location of the implant, bacterial contamination, delayed wound healing, surgical trauma, implant-related factors (type of implant system, implant surface), and others.
Smoking, occlusal overload, and other biological and biomechanical factors have also been noted to compromise implant success.8,9 More recent studies have concluded that another significant factor of implant success is soft tissue thickness (or biotype).10-13 Some studies name lack of adequate keratinized tissue or attached mucosa among contributing factors of implant failure.14-16 This topic is controversial, and more studies are needed to prove or disprove its validity.
Tension problem — alarming trend
This article focuses on an alarming issue of implants loosing support and integration due to excessive soft tissue pull or tension from a shallow vestibule and/or dense frenum pull. We now have an abundant number of cases that exhibit tissue recession and possibly late loss failure. For example, Figures 1A-1C show a case of late implant failure. In 1996, the patient received an implant on tooth No. 8. The immediate result was very good, both functionally and esthetically (Figure 1A). However, the 2007 follow-up visit revealed severe gingival recession (Figure 1B depicts the recession and distinct frenum pulls). The cross-sectional image from 2007 (Figure 1C) demonstrates the lack of bone in the facial and apical aspects of the implant. The presence of frenum pulls (evident in Figure 1A from 1996) should have been a reason for concern. We believe that a prophylactic release of the frenum pull prior to implant loading would have prevented such outcome. Today, we would have addressed the frenum pull with a CO2 frenectomy prior to implant loading.
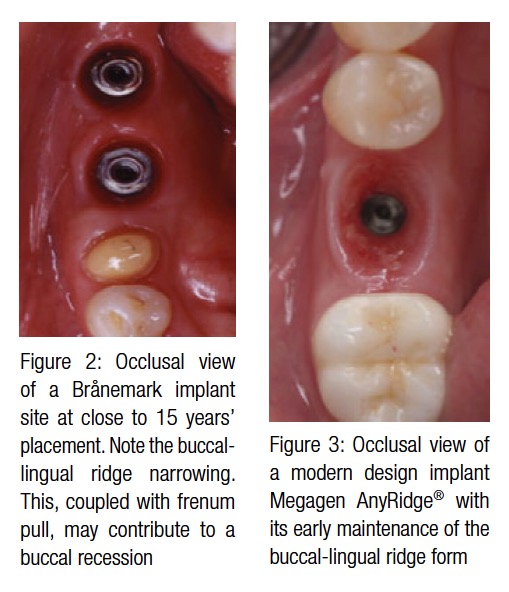
Figure 2 demonstrates two single Brånemark implants placed nearly 15 years prior. Natural buccal and lingual narrowing of the alveolar ridge and high mucogingival junction combined with frenum pull could possibly contribute to the buccal gingival recession and, consequently, the implant failure. In this case, the patient could have benefited from the tissue tension release.
Figure 3 presents the occlusal view of a modern design Megagen AnyRidge® implant with its early maintenance of the buccolingual ridge form. In other words, the buccal frenum pull was prophylactically released to alleviate tissue tension and lower the risk of marginal tissue recession in the future. Figure 3 clearly demonstrates healthy gingival mucosa in the absence of any frenum pulls or tension.
This article identifies the tissue tension problem and demonstrates a quick, effective, and relatively painless concept to prophy-lactically address this issue with the adjunctive use of a 10.6 micron SuperPulse CO2 laser.
Sources of pull and tension
High muscle attachment: Sometimes, muscle attachment extends onto the crest of the ridge, which results in a shallow vestibule. Such muscle attachment can create space inadequate for a substantial implant-supported denture (or any denture, for that matter). In addition, muscle fibers exert considerable tension on peri-implant mucosa, which may eventually result in gingival recession. In such cases, a simple vestibular extension procedure can solve the problem. Small penetration depth of the CO2 laser is important for the vestibular extension procedure because it affords the clinician precise control over the depth of incision.
Frenum pull: Frena are folds of mucous membrane containing fibrous connective tissue that attach lips and cheeks to the alveolar mucosa, the gingiva, and the underlying periosteum.17 Some frenal attachments can be dense, pulling on the attached gingiva. Tissue tension caused by the presence of frenum pull can be an important etiological factor in progressive gingival recession around posts and in eventual strut exposure.18,19 In the case of implant-retained denture prosthesis, frenum can limit denture extensions and even affect the seal and retention of the denture.18,20 The CO2 laser frenectomy procedure that releases tension exerted by the frenum pull creates a better chance of long-term success for a dental implant without sutures pulling, less postoperative swelling, and only minor pain or discomfort.
Lack of keratinized mucosa or attached gingival mucosa: Another risk factor is lack of attached gingiva. The width of attached gingival mucosa varies for different patients and even for different teeth in the same patient. In the oral cavity, attached gingiva (keratinized masticatory mucosa) meets movable alveolar mucosa (lining mucosa) at the mucogingival junction (MGJ). Clinically, the MGJ is identified by a mucogingival groove and the change from the pale pink of the attached gingiva to the bright pink of the movable alveolar mucosa.20 Width of keratinized mucosa is the distance between the mucogingival junction (MGJ) and the coronal aspect of the keratinized mucosa.14 There is no unequivocal consensus on the role the presence of keratinized mucosa plays in maintaining implant health.21-23 Literature suggests that a greater width of keratinized mucosa is advantageous due to the following:
- It provides a resistant barrier to plaque-induced inflammation
- It replaces non-keratinized margins to prevent recession
- It deepens vestibules to provide better access for tooth brushing
- It dissipates functional and masticatory stress placed on the gingival margin of a restoration
- It facilitates oral hygiene, and improves esthetics and patient comfort14,16,22
Clinicians generally agree about the link between the insufficient amount of keratinized mucosa or attached gingiva and marginal tissue recession. A study by Chung, et al.,14 has found that dental implants with insufficient attached gingiva show more plaque accumulation and mucosal inflammation than implants with adequate attached gingiva. If attached gingiva is insufficient (less than 4 mm), and the MGJ is positioned high, mucosa surrounding the implant is mobile and easily retractable during mastication and speech.24 Such tissue retraction can facilitate the introduction of plaque into the peri-implant pocket14,24 and lead to gingival recession. The presence of adequate (approximately 4 mm) attached gingiva correlates with mucosal health and can help prevent inflammation in peri-implant tissues. Al-Sabbagh and Bhavsar24 pointed out that wide zone of attached peri-implant gingiva provides better gingival seal around the implant. Another study has shown the correlation between lack of attached gingiva and crestal bone loss of 2 mm or more.25 These findings lead many clinicians, the authors included, to believe that the creation of sufficient amount of attached gingiva around implants is important and can potentially prevent implant failure. Width of the attached gingiva may be increased by a local vestibuloplasty.
To summarize, tissue tension or pull due to a shallow vestibule (caused by high muscle attachment), dense frenum, or lack of keratinized/attached mucosa can contribute to gingival recession. In addition to causing an esthetic problem, mucosal recession that denudes threads or a rough implant surface might impede the ability of the patient to maintain the implant clean from plaque. The resulting inflammation and infection create the risk of potential peri-implant bone loss and eventual implant failure.26,27
Proposed solution
In order to effectively release tension created either by a high muscle attachment and/or dense frenum, or high mucogingival junction with only a small amount of gingival mucosa, we recommend performing a CO2 laser frenectomy and/or vestibuloplasty with secondary epithelialization.
Why CO2 laser?
Not all lasers are equally efficient at both tissue vaporization (i.e., ablation or cutting) and coagulation. The difference is illustrated in the absorption spectra for main soft tissue chromophores28,29 in Figure 4. Some dental laser wavelengths (around 3,000 nm, such as Erbium lasers) are well absorbed by the water-rich soft tissue and are great at cutting but are not as efficient at coagulating.29 Other dental laser wavelengths (around 1,000 nm, such as diodes and Nd:YAG) are efficient coagulators, but inefficient scalpels30 since they are poorly absorbed by the soft tissue.
The 10,600 nm CO2 laser wavelength is efficient at both vaporizing and coagulating the soft tissue simultaneously (Figure 4), although it is not as good as Erbium laser at cutting and not as good as diode/Nd:YAG at coagulating. Most importantly, the CO2 laser’s coagulation depth closely matches the blood capillary diameters,29 as illustrated in Figure 5.

Laser pulsing is also as important for laser surgery as the wavelength: the short and powerful pulses are often superior to long and weak ones. The exact physics of pulsed laser surgery deals with the Thermal Relaxation Time,29,30 which depends both on tissue’s light absorption and tissue’s thermal diffusivity, first described by Einstein32. The irradiated tissue is ablated (vaporized) the most efficiently when the pulse duration is much shorter than the Thermal Relaxation Time. The tissue adjacent to the ablated zone cools down most efficiently when the length of time between laser pulses significantly exceeds the Thermal Relaxation Time. Such laser pulsing, referred to as SuperPulse, minimizes the depth of coagulation and is a must-have feature of any state-of-the-art soft tissue surgical CO2 laser.29
The optimal combination of the CO2 laser wavelength and pulsing allows for a char-free and bloodless surgery. This also allows for a scar-free, uncomplicated healing that is valued by surgeons across all specialties in dentistry, OMFS, ENT, plastic surgery, etc.
Laser beam spot size
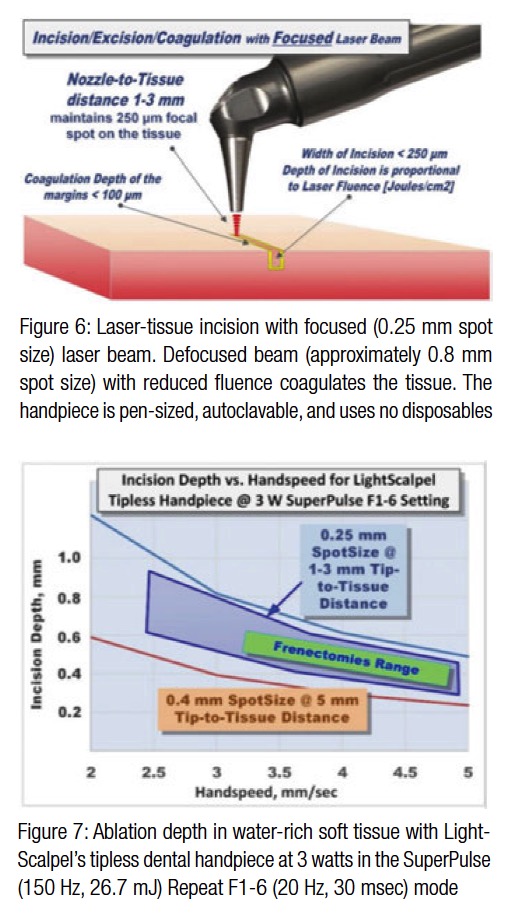 Just like the sharpness of the steel blade defines the quality and ease of the incision, the size of the laser beam focal spot determines the quality of the laser cut. The smaller (or sharper) the focal spot of the beam, the narrower and deeper the incision. Just like a dull blade cannot produce a quality incision, an oversized laser beam spot cannot produce a precise and narrow incision. For cutting, the LightScalpel laser handpiece is maintained 1-3 mm away from the tissue and is moved at a hand speed of a few millimeters per second — as illustrated in Figure 6. For a rapid switch from cutting to just photo-coagulation, the laser beam can be defocused. Defocusing can be achieved either by selecting a larger spot size, or by simply moving the handpiece away from the tissue (by approximately 8 mm for LightScalpel tipless laser handpieces), and “painting” the “bleeder” for enhanced hemostasis (Figure 6).
Just like the sharpness of the steel blade defines the quality and ease of the incision, the size of the laser beam focal spot determines the quality of the laser cut. The smaller (or sharper) the focal spot of the beam, the narrower and deeper the incision. Just like a dull blade cannot produce a quality incision, an oversized laser beam spot cannot produce a precise and narrow incision. For cutting, the LightScalpel laser handpiece is maintained 1-3 mm away from the tissue and is moved at a hand speed of a few millimeters per second — as illustrated in Figure 6. For a rapid switch from cutting to just photo-coagulation, the laser beam can be defocused. Defocusing can be achieved either by selecting a larger spot size, or by simply moving the handpiece away from the tissue (by approximately 8 mm for LightScalpel tipless laser handpieces), and “painting” the “bleeder” for enhanced hemostasis (Figure 6).
Laser power density and depth of incision
For a laser scalpel, the power density of the focused laser beam is equivalent to the mechanical pressure that is applied to a cold steel blade. In other words, greater laser fluence29 (i.e., greater power density and slower hand speed) results in greater depth and rate of soft tissue removal. During each SuperPulse pulse, the ablation depth δ is given by the formula δ = A (E – Eth) / Eth for the steady state ablation conditions,30 where A is the absorption depth from Figure 4 and Eth is the ablation threshold fluence,29 and E is the fluence during the SuperPulse pulse. At the 10.6 µm wavelength of the CO2 laser, the ablation threshold for a water-rich soft tissue with an assumed water content of 75% equals approximately Eth = 3 J/cm2. For repetitive pulses that are scanned across the soft tissue, the fluence is defined by the pulse frequency and the hand speed: i.e., the depth of incision depends on laser power settings, spot size, and the surgeon’s hand speed33,34 (Figure 7).
Uses of CO2 lasers in implant dentistry
CO2 lasers have been used and studied in many areas of implant dentistry.18,33-39 Some authors consider the newer CO2 lasers the most versatile of all the soft tissue lasers in implant dentistry.38 For example, the CO2 laser is effective for creating flaps, incisions for a sinus lift, stage II implant uncovering, treatment of peri-implantitis, removal of gingival hyperplasia, epulis, fibromas, graft donor site hemostasis, and so on. The CO2 laser allows the clinician to address such critical aspects of implant therapy as the extraction site sterilization, excess cement removal, troughing for digital impression, and muscle pull release. All of the above is important for long-term success of implants.
Hemostasis: The CO2 laser’s excellent hemostasis and coagulation (due to close match between coagulation depth and gingival blood vessel diameters) allows to perform surgery even in the most vascularized areas. It affords the clinician improved visibility of the surgical field, and therefore, allows for more precise and accurate tissue removal.40 Due to the efficient hemostasis, intraoral surgical wounds often do not require suturing or surgical dressing and can be left to heal by secondary intention.18,41
Minimal post-operative swelling: Another advantage of the CO2 laser is minimal postoperative swelling and edema due to the intraoperative closure of lymphatic vessels on the margins of the CO2 laser incision. Lymphatic vessels regenerate in approximately 8 to 10 days after capillary-vessel proliferation.42
Reduced post-operative pain and discomfort: Although it is generally difficult to evaluate pain, less discomfort was reported with the CO2 laser surgery than with conventional one.43 In the study by Niccoli-Filho, et al.,44 the patients reported minimal discomfort only during the first 24 hours after the CO2 laser surgery. Haytac and Ozcelic45 reviewed the use of the CO2 laser in frenectomies. Based on patient pain perceptions during this procedure, they concluded that the laser treatment was less painful than the one performed conventionally, with a scalpel. In Neckel’s study46, vestibuloplasty was performed on 40 patients with either a conventional blade or with a CO2 laser. Both groups showed similar increase in the vestibular height, but patients in the CO2 laser group reported less pain and discomfort. Strauss, et al.,47 and Deppe, et al.,48 compared the recovery process following CO2 laser surgery with that following cryosurgery and electrosurgery and reported that with the CO2 laser healing was faster and less painful.
Laser healing and reduced scarring
Significantly reduced wound contraction and scarring are among the most important advantages of CO2 laser treatment.41,49-51 In CO2 laser-irradiated wounds, the healing process is characterized by a more prominent fibroblastic proliferation, with young fibroblasts actively producing collagen. Several studies51-53 found that in comparison with scalpel wounds, only a small number of myofibroblasts (cells responsible for wound contraction) are present in the CO2 laser-excised wounds. Seventy-two hours after the CO2 laser surgery, a fibroserous membrane forms over the wound to replace the superficial necrotic layer of the laser-irradiated site.54,55 Approximately 2 weeks postoperatively, the wound starts to epithelialize from the periphery toward the center. The epithelial covering of the laser wound is thinner and parakeratotic in comparison with the epithelium that forms after scalpel resection. This could account for the superb esthetic outcome of CO2 laser surgery with smooth pliable new tissue and no fibrosis or scarring, while a scalpel can leave some scarring.50 Decreased wound contraction combined with minimal lateral tissue damage, less traumatic surgery, precise control over the depth of incision, and excellent hemostatic ability make the CO2 laser a safe and efficient alternative to a conventional scalpel.
To summarize, the physics of laser-tissue interaction, the considerable amount of peer-reviewed literature on uses of the CO2 laser in implant dentistry, and our own clinical experience confirm our choice of the CO2 laser as the surgical tool for soft tissue tension release, frenectomies, and vestibular extensions.
Case study 1
A 75-year-old female patient presented for recurrent caries in the lower left premolars Nos. 20 and 21, underneath crowns (Figures 8A-8B). In addition, the teeth had weak coronal structure. Since they deemed unstable for long-term survival, it was decided to extract them and replace them with two single implants. Six weeks after extraction, the patient returned for a flapless implant placement (Figures 9-11B).
Six weeks after implant placement (the healing phase), the patient came in for the implant crowns loading. Figures 12A and 12B demonstrate healthy appearance of peri-implant mucosa. However, horizontal rotation of the labial and buccal tissue revealed tension created by the movable mucosa very close to the implants. This was due to the narrow zone of attached gingival mucosa (this narrow zone is especially noticeable in Figures 10B, 11A, and 12A. The pale pink tissue is the attached gingiva, whereas the bright pink is the movable alveolar mucosa).

It became apparent that the encroaching movable mucosa and the close buccal frenum insertion (clearly seen in Figure 8A), both exerted tension on the peri-implant tissue, especially noticeable when lip or cheek were manipulated. This created potential for tissue recession and could eventually facilitate the implants’ failure. It was decided to perform a CO2 laser frenectomy/vestibuloplasty to alleviate the tension and possibly increase the width of attached gingiva.
Surgical Laser Equipment: A flexible-fiber dental CO2 laser LightScalpel LS-1005 was utilized with a dental angled tipless handpiece with a 0.25-mm focal spot diameter
Laser Settings: 3 watts; SuperPulse Repeat Pulse Mode F1-6
Anesthesia: No local anesthesia was administered; only topical was used. (With some patients local anesthesia is given.)
CO2 laser procedure: Traction was applied labially and buccally to expose the tissue tension between the keratinized gingival mucosa and movable alveolar mucosa as well as the frenum pull. Importantly, maintaining traction significantly facilitates laser cutting.
A horizontal CO2 laser incision was made along the mucogingival junction (or the line where the tension is most apparent) following the contours of the underlying bone. The tip of the laser handpiece was held perpendicular to the target tissue at a distance of 1 mm–2 mm from it. The handpiece was moved at the recommended speed of 4-5 mm/second. While making the laser incision, the clinician feels the release of the tissue tension. If the created incision, however, does not provide satisfactory tension relief, additional passes may be needed. Typically, between 4 and 8 laser passes are made to achieve the desired depth of incision, and the procedure usually takes under 1 minute.

Normally, the CO2 laser produces excellent visualization and a clear operatory field (Figure 12C). If, however, slight bleeding occurs after the incision is made, the laser beam is de-
focused by increasing the nozzle-to-tissue distance to quickly obtain hemostasis.
Wound closure: No suturing was used after this procedure. The wound was left to heal by secondary intension. Generally, the wound re-epithelializes within 2.5-3 weeks.
Postoperative instructions: The patient was released from the clinic with instructions to do warm salt water rinses 4 times a day and to apply topical antibiotic and vitamin E gel twice daily directly to the area. The patient was advised to avoid spicy, acidic, or harsh foods with sharp edges, or caustic mouth rinses. She was educated about the maintenance of oral hygiene. The patient reported to be completely pain-free 24 hours after the surgery.
Follow-up examination: Two weeks after the procedure, the patient returned for a check-up. Healing progressed well. No signs of swelling or inflammation were noted. The patient did not express any complaints during the postoperative period. The 4-week follow-up visit showed beautifully healed tissue with no scarring (Figures 13A and 13B). The recovery was uneventful. Figures 14A and 14B present the buccal final view of two implant crowns in place and stable soft tissue. Traction applied coronally with a perio probe demonstrates the lack of frenum pull or mucosal tension at the site (compare with pre-vestibuloplasty view in Figure 11A).
Case study 2
 The 73-year-old female patient had a single implant on tooth No. 29 placed. Figures 15A and 15B show a noticeable buccal frenum pull present in the region. High mucogingival junction (evident in Figure 15B) indicates insufficient amount of keratinized attached gingiva. It was decided to perform a CO2 laser frenectomy/vestibuloplasty to increase the width of the attached gingiva. The laser procedure was performed utilizing the same laser settings and following the same protocol as previously described in Case study 1:
The 73-year-old female patient had a single implant on tooth No. 29 placed. Figures 15A and 15B show a noticeable buccal frenum pull present in the region. High mucogingival junction (evident in Figure 15B) indicates insufficient amount of keratinized attached gingiva. It was decided to perform a CO2 laser frenectomy/vestibuloplasty to increase the width of the attached gingiva. The laser procedure was performed utilizing the same laser settings and following the same protocol as previously described in Case study 1:
- Topical or local anesthesia is administered.
- Traction to the lip or cheek is applied and maintained throughout the procedure.
- Laser handpiece is directed at 90º to the target tissue.
- Laser-tissue distance is kept at 1 mm-2 mm for an incision and 3 mm-4 mm for coagulation.
- Hand speed is maintained at 4-5 mm/second.
- An incision is made at the point where tissue tension is most apparent.
- The incision should follow the contours of the underlying bone.
- The incision is extended to the desired depth.
- No sutures are required.
- Patient is released from the clinic immediately after the procedure with instructions on how to maintain oral hygiene, to avoid acidic, caustic or harsh foods, drinks or mouth rinses. We recommend warm salt rinses 4 times a day; NSAIDs, if needed, topical antibiotic and vitamin E gel twice daily.
 Figure 16 demonstrates immediate postoperative view of the CO2 laser frenectomy/vestibuloplasty completed in less than 1 minute, bloodlessly, and without local anesthesia.
Figure 16 demonstrates immediate postoperative view of the CO2 laser frenectomy/vestibuloplasty completed in less than 1 minute, bloodlessly, and without local anesthesia.
Follow-up evaluation: At 4 weeks, the surgical site appeared completely healed (Figure 17). The patient gained 3 mm-4 mm of the vestibule depth, and although the frenum was still present, its attachment moved apically and further away from the implant (Figure 17).
Conclusion
The CO2 laser was chosen for this procedure for several reasons, such as the laser’s ability to provide instant hemostasis with a minuscule zone of thermal injury (sub 50 microns),31 lack of need for sutures, reduced wound contraction, smooth healing, and only minor pain and discomfort reported by patients.
The CO2 laser frenectomy/vestibuloplasty is a simple, neat, and quick procedure with minimal postoperative pain or discomfort for the patient. While by no means a panacea against implant failure, this procedure allows the clinician to remove one of the many risk factors — i.e., tissue tension, that can potentially compromise the long-term success of dental implants. Due to the above and based on years of clinical experience, the authors recommend the CO2 laser frenectomy/vestibuloplasty as an effective surgical solution.
Acknowledgment
Authors greatly appreciate the support and contribution from Anna (Anya) Glazkova, PhD, in preparing this material for publication.
- Albrektsson T, Zarb G, Worthington P, Eriksson AR. The long-term efficacy of currently used dental implants: a review and proposed criteria of success. Int J Oral Maxillofac Implants. 1986;1(1):11-25.
- Brånemark PI, Hansson BO, Adell R, Breine U, Lindström J, Hallén O, Ohman A. Osseointegrated implants in the treatment of the edentulous jaw. Experience from a 10-year period. Scand J Plast Reconstr Surg Suppl. 1977;16:1-132.
- Smith DE, Zarb GA. Criteria for success of osseointegrated endosseous implants. J Prosthet Dent. 1989;62(5):567–572.
- Proskin HM, Jeffcoat RL, Catlin A, Campbell J, Jeffcoat MK. A metaanalytic approach to determine the state of the science on implant dentistry. Int J Oral Maxillofac Implants. 2007;22 suppl:11-18.
- Misch CE, Perel ML, Wang HL, Sammartino G, Galindo-Moreno P, Trisi P, Steigmann M, Rebaudi A, Palti A, Pikos MA, Schwartz-Arad D, Choukroun J, Gutierrez-Perez JL, Marenzi G, Valavanis DK. Implant success, survival, and failure: the International Congress of Oral Implantologists (ICOI) Pisa Consensus Conference. Implant Dent. 2008;17(1):5-15.
- Heydenrijk K, Meijer HJ, van der Reijden WA, Raghoebar GM, Vissink A, Stegenga B. Microbiota around root-form endosseous implants: a review of the literature. Int J Oral Maxillofac Implants. 2002;17(6):829–838.
- Moy PK, Medina D, Shetty V, Aghaloo TL. Dental implant failure rates and associated risk factors. Int J Oral Maxillofac Implants. 2005;20(4):569–577.
- Steigenga J, Al-Shammari K, Misch C, Nociti FH Jr, Wang HL. Effects of implant thread geometry on percentage of osseointegration and resistance to reverse torque in the tibia of rabbits. J Periodontol. 2004;75(9):1233-1241.
- Kim Y, Oh TJ, Misch CE, Wang HL. Occlusal considerations in implant therapy: clinical guidelines with biomechanical rationale. Clin Oral Implants Res. 2005;16(1):26-35.
- Evans CD, Chen ST. Esthetic outcomes of immediate implant placements. Clin Oral Implants Res. 2008;19(1):73-80.
- Jung RE, Sailer I, Hammerle CH, Attin T, Schmidlin P. In vitro color changes of soft tissues caused by restorative materials. Int J Periodontics Restorative Dent. 2007;27(3):251-257.
- Levine RA, Huynh-Ba G, Cochran DL. Soft tissue augmentation procedures for mucogingival defects in esthetic sites. Int J Oral Maxillofac Implants. 2014;29 Suppl:155-185.
- Lee A, Fu JH, Wang HL. Soft tissue biotype affects implant success. Implant Dent. 2011;20(3):e38-47. doi: 10.1097/ ID.0b013e3182181d3d.
- Chung DM, Oh TJ, Shotwell JL, Misch CE, Wang HL. Significance of keratinized mucosa in maintenance of dental implants with different surfaces. J Periodontol. 2006;77(8):1410–1420.
- Bengazi F, Wennström JL, Lekholm U. Recession of the soft tissue margin at oral implants. A 2-year longitudinal prospective study. Clin Oral Implants Res. 1996;7(4):303-310.
- Greenstein G, Cavallaro J. The clinical significance of keratinized gingiva around dental implants. Compend Contin Educ Dent. 201;32(8):24-31;quiz 32, 34.
- Devishree SKG, Shubhashini PV. Frenectomy: A Review with the Reports of Surgical Techniques. J Clin Diagn Res. 2012 Nov; 6(9): 1587–1592. Published online 2012 Nov 15. doi: 10.7860/ JCDR/2012/4089.2572
- Namour S. Atlas of Current Oral Laser Surgery. Boca Raton, FL:Universal Publishers;2011.
- Weber SP. A mucobuccal fold extension for implant and other surgical procedures. J Prosthet Dent. 1972;27(4):423-433.
- Nanci A. Oral mucosa. In: Nanci A, ed. Ten cate’s oral histology: development, structure, and function. 7th ed. St.Louis, MO:Mosby;2008:319-357.
- Cairo F, Pagliaro U, Nieri M. Soft tissue management at implant sites. J Clin Periodontol. 2008;35(8 suppl):163-167.
- Bouri A Jr, Bissada N, Al-Zahrani MS, Faddoul F, Nouneh I. Width of keratinized gingiva and the health status of the supporting tissues around dental implants. Int J Oral Maxillofac Implants. 2008;23(2):323-326.
- Yeung SC. Biological basis for soft tissue management in implant dentistry. Aust Dent J. 2008;53(suppl 1):S39-S42.
- Al-Sabbagh M, Bhavsar I. Key local and surgical factors related to implant failure. Dent Clin North Am. 2015;59(1):1-23. doi: 10.1016/j. cden.2014.09.001.
- Block MS, Kent JN. Factors associated with soft- and hard-tissue compromise of endosseous implants. J Oral Maxillofac Surg. 1990;48(11):1153–1160.
- Albrektsson T, Dahlin C, Jemt T, Sennerby L, Turri A, Wennerberg A. Is marginal bone loss around oral implants the result of a provoked foreign body reaction? Clin Implant Dent Relat Res. 2014;16(2):155- 165. doi: 10.1111/cid.12142. Epub 2013 Sep 4.
- Dibart S, Warbington M, Su MF, Skobe Z. In vitro evaluation of the implant-abutment bacterial seal: the locking taper system. Int J Oral Maxillofac Implants. 2005;20(5):732-737.
- Jacques SL. Optical properties of biological tissues: a review. Phys Med Biol. 2013;58(11):R37-61.
- Vitruk P. Oral soft tissue laser ablative and coagulative efficiencies spectra. Implant Practice US. 2014;7(6):22-27.
- Vogel A, Venugopalan V. Mechanisms of pulsed laser ablation of biological tissues. Chem Rev. 2003;103(2):577-644.
- Wilder-Smith P, Arrastia AM, Liaw LH, Berns M. Incision properties and thermal effects of three CO2 lasers in soft tissue. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 1995; 79(6):685-691.
- Einstein A. Über die von der molekularkinetischen theorie der wärme geforderte bewegung von in ruhenden flüssigkeiten suspendierten teilchen (translation: On the movement of small particles suspended in a stationary liquid demanded by the molecular-kinetic theory of heat). Ann Phys. 1905;2(Doc. 16):549-560.
- Linden E, Vitruk P. SuperPulse 10.6 um CO2 laser-assisted, closed flap treatment of peri-implantitis. Implant Practice US. 2015: 8(4):30-34.
- Cobb C, Vitruk P. Effectiveness of a super pulsed CO2 laser for removal of biofilm from three different types of implant surfaces: an in vitro study. Implant Practice US. 2015;8(3):20-28.
- Romanos G. Laser surgical tools in implant dentistry for long-time prognosis of oral implants. Int Congress Series. 2003;1248:111.
- Deppe H, Horch H, Helmut G, Brill T, Wagenpfeil S, Donath, K. Peri-implant care with the CO2 laser: in vitro and in vivo results. Med Laser Appl. 2005;20:61-70.
- Deppe H, Horch H. Laser applications in oral surgery and implant dentistry. Lasers Med Sci. 2007;22:217-221.
- Julian J. Lasers in implant dentistry. In: Convissar RA. Principles and Practices of Laser Dentistry. St. Louis, MO: Mosby; 2011:114-38.
- Romanos G, Ko HH, Froum S, Tarnow D. The Use of CO2 Laser in the Treatment of Peri-implantitis. Photomed Laser Surg. 2009;27(3):381-386.
- Kotlow LA. Lasers in pediatric dentistry. Dent Clin North Am. 2004;48(4):889-922.
- Zaffe D, Vitale MC, Martignone A, Scarpelli F, Botticelli AR. Morphological histochemical and immunocytochemical study of CO2 and Er:YAG laser effect on oral soft tissues. Photomed Laser Surg. 2004;22(3):185-189.
- Lambrecht JT, Stübinger S, Hodel Y. Treatment of intraoral hemangiomas with the CO2 laser. J Oral Laser Appl. 2004;4:89-96.
- Pogrel MA. The carbon dioxide laser in soft tissue preprosthetic surgery. J Prosthet Dent. 1989;61(2):203-208.
- Niccoli-Filho W, Neves ACC, Penna LAP, Seraidarian PI, Riva R. Removal of epulis fissuratum associated to vestibuloplasty with carbon dioxide laser. Lasers in Med Sci. 1999;14(3):203–206.
- Haytac MC, Ozcelik O. Evaluation of patient perceptions after frenectomy operations: a comparison of carbon dioxide laser and scalpel techniques. J Periodontol. 2006;77(11):1815-1819.
- Neckel CP. Vestibuloplasty: a retrospective study on conventional and laser operation techniques. Lasers in Dentistry. 1999;76(5)18–23.
- Strauss RA, Fallon SD. Lasers in contemporary oral and maxillofacial surgery. Dent Clin North Am. 2004;48(4):861-888.
- Deppe H, Horch HH. Current status of laser applications in oral and cranio-maxillofacial surgery. Med Laser Appl. 2007;22(1):39-42.
- Zeinoun T, Nammour S, Dourov N, Aftimos G, Luomanen M. Myofibroblasts in healing laser excision wounds. Lasers Surg Med. 2001;28(1):74-79.
- Wang X, Ishizaki NT, Matsumoto K. Healing process of skin after CO2 laser ablation at low radiance: a comparison of continuouswave and pulsed mode. Photomed Laser Surg. 2005;23(1):20-26.
- Grbavac RA, Veeck EB, Bernard JP, Ramalho LM, Pinheiro AL. Effects of laser therapy in CO2 laser wounds in rats. Photomed Laser Surg. 2006;24(3):389-396.
- de Freitas AC, Pinheiro AL, de Oliveira MG, Ramalho LM. Assessment of the behavior of myofibroblasts on scalpel and CO2 laser wounds: an immunohistochemical study in rats. J Clin Laser Med Surg. 2002;20(4):221-225.
- Fisher SE, Frame JW, Browne RM, Tranter RM. A comparative histological study of wound healing following CO2 laser and conventional surgical excision of canine buccal mucosa. Arch Oral Biol. 1983;28(4):287-291.
- Basu MK, Frame JW, Rhys Evans PH. Wound healing following partial glossectomy using the CO2 laser, diathermy and scalpel: a histological study in rats. J Laryngol Otol. 1988;102(4):322-327.
- Tambuwala A, Sangle A, Khan A, Sayed A. Excision of oral leukoplakia by CO2 lasers versus traditional scalpel: A comparative study. J Maxillofac Oral Surg. 2014;13(3):320-327.
Stay Relevant With Implant Practice US
Join our email list for CE courses and webinars, articles and mores

 Jack T. Krauser, DMD, is a well-known dental implant surgeon and periodontist. His accomplishments include designing systems of implants and prosthetic parts that are currently used in the marketplace. Dr. Krauser has lectured frequently, contributed to implant-related texts, co-authored a recently published book Dental implants: The Art and the Science, and has over 20 journal articles in peer-reviewed journals. Dr. Krauser’s clinical cases have been included in the only ADA review of implant systems in evaluation. Dr. Krauser is accredited with the AAP, ICOI, and AO. He lectures around the world on 3D planning for optimal implant positioning, implant complications, implant surface analysis, and hands-on placement. Dr. Krauser has held hospital appointments at Palm Beach Gardens Medical Center and West Boca Medical Center in Florida. He graduated from the University of Pennsylvania with a DMD degree and achieved his certification in Advanced Dental Standing, Periodontics from Boston University’s Goldman School of Graduate Dentistry.
Jack T. Krauser, DMD, is a well-known dental implant surgeon and periodontist. His accomplishments include designing systems of implants and prosthetic parts that are currently used in the marketplace. Dr. Krauser has lectured frequently, contributed to implant-related texts, co-authored a recently published book Dental implants: The Art and the Science, and has over 20 journal articles in peer-reviewed journals. Dr. Krauser’s clinical cases have been included in the only ADA review of implant systems in evaluation. Dr. Krauser is accredited with the AAP, ICOI, and AO. He lectures around the world on 3D planning for optimal implant positioning, implant complications, implant surface analysis, and hands-on placement. Dr. Krauser has held hospital appointments at Palm Beach Gardens Medical Center and West Boca Medical Center in Florida. He graduated from the University of Pennsylvania with a DMD degree and achieved his certification in Advanced Dental Standing, Periodontics from Boston University’s Goldman School of Graduate Dentistry. Peter Vitruk, PhD, MInstP, CPhys, is a founder of LightScalpel, LLC. He is a member of the Institute of Physics, United Kingdom, and is the member of the Science and Research Committee, Academy of Laser Dentistry, United States. He is also on the faculty of the California Implant Institute and Global Laser Oral Health, both in the United States. Dr. Vitruk can be reached at 1-866-589-2722 or pvitruk@lightscalpel.com.
Peter Vitruk, PhD, MInstP, CPhys, is a founder of LightScalpel, LLC. He is a member of the Institute of Physics, United Kingdom, and is the member of the Science and Research Committee, Academy of Laser Dentistry, United States. He is also on the faculty of the California Implant Institute and Global Laser Oral Health, both in the United States. Dr. Vitruk can be reached at 1-866-589-2722 or pvitruk@lightscalpel.com.

