Dr. Pierre J. Tedders discusses a technique for grafting ridges requiring large areas of reconstruction
Management of the comprehensive dental implant patient routinely requires guided-bone regenerative techniques. The benefits that the edentulous patient receives from an implant reconstruction can be life altering. Although depending on the specific patient, the duration of edentulism, the prosthetic, and utilization of the prosthetic, the alveolar and basal bone available for the reconstruction may range from sufficient to severe atrophy.
[userloggedin]
Beyond the intraoral effects, these patients also suffer consequences of esthetics, decreased function, and psych-ological effects. Numerous predictable surgical protocols have been developed to manage the atrophic ridge. In those patients who have sufficient vertical height, with moderate to severe horizontal deficiency, an onlay graft via “tenting” procedure may be most appropriate. This technique is useful in grafting ridges requiring large areas and may not result in the excessive facial remodeling of the implant as traditional block grafting.

The following case will demonstrate the surgical procedure to overcome these types of deficiencies. The surgical plan includes a full periosteal flap reflection, nasal mucosal reflection, augmentation of the alveolar ridge, via onlay grafting with tenting to gain necessary width, and augmentation of the nasal cavity to gain height for the anterior implants utilizing Plasma Rich in Growth Factors (PRGF). The healing phase for this patient was 3 months prior to placing six root-form endosseous implants. This healing phase timing is appropriate when combining growth factors and the well-documented initial stability of the OCO Biomedical Engage™ implant with its Bull Nose Auger™ tip design.
Clinical case
The patient presented with a chief complaint of ill-fitting maxillary complete denture. She reported that the fit and function of her maxillary prosthesis has negatively affected the quality of her life. The patient is ASA I with no contraindications to dento-alveolar surgery. Thorough clinical and radiographic examination reveals moderate to severe maxillary arch deficiency (Figures 1-3).
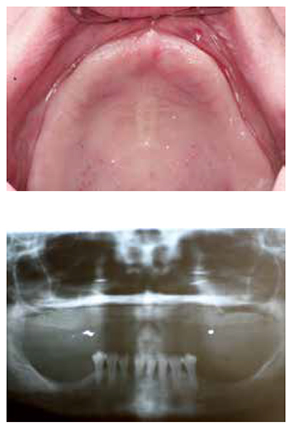
Consideration was made for a traditional block-grafting procedure and combined block grafting with ridge splitting. Due to the size of the proposed graft and current known limitations to block grafting, the option was made for onlay grafting via a tenting procedure with sub-nasal elevation and augmentation with PRGF.
A full thickness incision was made maintaining 3 mm of keratinized tissue to the facial of the incision line. Posterior superior vertical releasing incisions were made, and a full thickness reflection was completed exposing the facial alveolar and basal bone exposing the extent of the deficiency (Figure 4).
Minimal alveoloplasty was completed followed by decortication, for regional acceleratory phenomenon aiding in angiogenesis (Figure 5), and bilateral reflection of the sub-nasal mucosa. Care is taken not to reflect the nasal mucosa as far posteriorly as the nasal cavity extends (reflection approximately 8-10 mm posteriorly) (Figure 6).


Bone fixation screws (Salvin® Dental) were strategically fixed to the maxilla. The fixation screws serve to tent the collagen type I membrane and minimize the negative effects of compression from the muco-periosteum, peri-oral soft tissues, and musculature (Figures 7 and 8). The fixation screws are approximated to slightly larger than the new proposed alveolar ridge — the final result ending with a minimum of 2 mm of bone to the facial and palatal of the dental implant.
Approximately 64 cc of blood was drawn from the right antecubital fossa and prepped with 8 cc allograft (250 um-1000 um) for PRGF separated into “membrane” (Figure 10) and “graft” fractions. The allograft is osteoconductive, or serves as a scaffold for the predictable inflammatory response and remodeling. The PRGF increases the volume of localized growth factors, TGF-beta, and PDGF “inflammatory cytokines.” The growth factors accelerate the inflammatory cascade, the rate of angiogenesis, and further serves as a filler. With the addition of growth factors, there is a significant decrease in reported postoperative complications.
A type I collagen membrane was modified with tissue scissors and secured to the palatal tissue with 4-0 polyglycolic acid (PGA) sutures (Figure 11). The collagen membrane serves several functions: 1) the collagen membrane prevents epithelial migration into and displacing the graft and 2) contains the graft material.

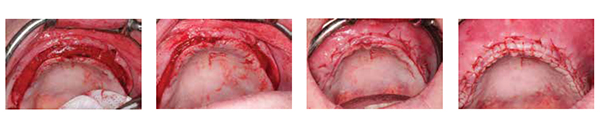
The PRGF and allograft “graft” were moderately condensed between the nasal floor and the nasal mucosa to a total height of approximately 15 mm from the alveolar crest. The graft was then moderately condensed to follow the new proposed facial-palatal ridge to slightly overfill beyond the tenting screws to form the proposed new facial surface of the maxillary alveolus (Figure 11). The collagen membrane was then folded over and secured to contain the graft (Figure 12).
With the entirety of the graft condensed and secured with the collagen type I membrane, the tissues do not approximate passively. The number one complication of a ridge reconstruction of width is suture line opening. Suture line opening exposes the underlying graft, and a negative inflammatory response follows, leading to excessive remodeling and potential infection (Figure 13).
Horizontal periosteal releasing incisions and blunt dissection of the tissues was completed to allow for the tissues to approximate passively (Figure 14). Initially, 12 horizontal mattress sutures were utilized to approximate the tissues. The closure must be primary and tension free. Placing the horizontal mattress sutures at the mucogingival junction everts the tissues and releases the tension off the incision line. Further, multiple sutures must be used to prevent suture line opening due to suture failure during the phase of edema (Figure 15).
Final incision line closure was completed with continuous interlocking suture with 4-0 PGA (Figure 16). The patient was prescribed a postoperative chemotherapeutic regimen, chlorhexidine rinse, an anti-inflammatory, and a narcotic pain reliever as needed. The patient was seen at 1 week, 2 weeks for suture removal (Figure 17), and 4 weeks to monitor the healing process.
The patient then entered a total 12-week healing phase. At the 12-week time frame, the new maxillary arch was exposed using a full-thickness incision from right to left tuberosity with disto-vertical releasing incisions to gain access to the tenting screws for removal and implant placement. The remaining ridge demonstrated sufficient bone regeneration, to the head of the fixation screw, for the planned six 4.0 x 12 OCO Engage™ implants (OCO Biomedical) (Figure 18).

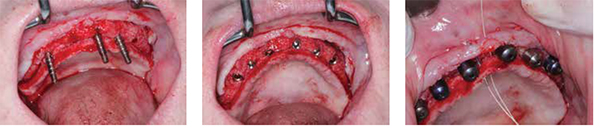

All tenting screws were removed, and minor alveoloplasty (Figure 19) was completed. Paralleling pins (Figure 20) were used to maintain parallelism for the placement of six 4.0 x 12 OCO Engage implants utilizing the simplified OCO Biomedical two-drill surgical sequence.

The surgery was performed in a single-stage approach, and 5-mm tall trans-mucosal healing abutments were placed. For this specific surgery, the OCO Engage implant was selected for its outstanding primary stability. The Engage is a bone-level implant with a variable self-taping thread pattern. The coronal micro threads increase the bone-to-implant surface contact, and combined with the Bull Nose Auger tip, provide the initial stability required for predictable success with a limited healing phase. The initial stability of the implants ranged from 40N/cm to 60N/cm of torque (sufficient for immediate load). The tissues were approximated with interrupted horizontal mattress sutures (Figures 21 and 22), and the patient was seen at the 2-week and 4-week postoperative visits. At the 3-month follow-up visit, the implants were torque tested to 35N/cm, and the patient was referred to her general dentist to complete the prosthetic phase of her treatment.
The final prosthesis was a maxillary implant-retained overdenture. Note: In treatment planning of the implant overdenture, if the palate is to be removed out of the final denture, the implants must be splinted. The palate and the alveolar ridge provide the support and stability of the prosthesis. If individual locators are utilized, and the palate is removed, the prosthetic becomes an implant-retained and implant-supported prosthesis, leading to potential progressive bone loss around the implants. All implants can be seen on the postoperative pan (Figure 23).
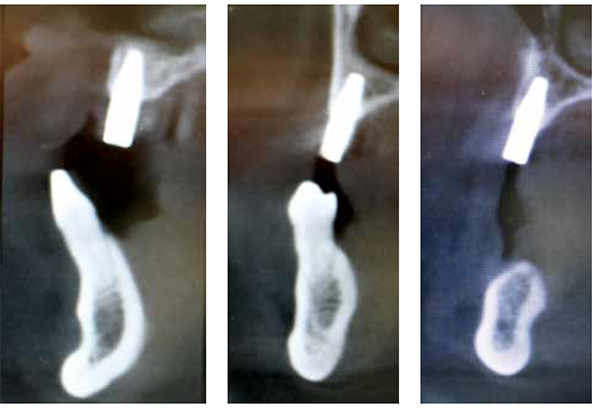
The patient presented for a 2-year postoperative evaluation on February 15, 2016. A clinical examination (Figure 24A) and CBCT were performed. All tissues remain firm, pink, and asymptomatic. The remaining maxillary ridge has maintained width and bone to the facial of the implants. (Note comparison photos preoperative Figure 24B and post-operative.) Figures 25C-25D demonstrate the augmented bone of the nasal cavity. Figure 25 shows the 2-year postoperative CBCT.
Thorough knowledge and experience with the different ridge augmentation and bone manipulation techniques offer reconstructive options that directly improve the quality of life for our patient base. This case presented an onlay graft via a tenting procedure, which offers another option to obtain necessary bone quantity for implant placement. The procedure is relatively simple and may offer more predictable results to traditional block grafting.
The patient is continuing her treatment with an immediate placement and immediate load transitional mandibular hybrid denture with five mandibular OCO implants in the A, B, C, D, and E positions in March 2016. Additionally, it has been highly recommended that an implant-retained and tissue-supported maxillary implant overdenture with a locator bar be fabricated.
The patient stated that there has been a significant improvement in the quality of her life following her implant reconstruction. This was, and should be, the ultimate goal of our implant treatments.

[/userloggedin]
[userloggedout][/userloggedout]
Stay Relevant With Implant Practice US
Join our email list for CE courses and webinars, articles and mores

 Pierre J. Tedders, DDS, graduated from the University of Detroit-Mercy 1999 and received additional training at the University of Michigan/ VA Hospital general practice residency. He is a graduate of the Medical College of Georgia Maxi Course. Dr. Tedders has limited his practice to surgical and restorative dental implantology in southern and central Michigan. He is a Diplomate of the American Board of Oral Implantology/Implant Dentistry, an Associate Fellow of the American Academy of Implant Dentistry, and Educator for OCO Biomedical.
Pierre J. Tedders, DDS, graduated from the University of Detroit-Mercy 1999 and received additional training at the University of Michigan/ VA Hospital general practice residency. He is a graduate of the Medical College of Georgia Maxi Course. Dr. Tedders has limited his practice to surgical and restorative dental implantology in southern and central Michigan. He is a Diplomate of the American Board of Oral Implantology/Implant Dentistry, an Associate Fellow of the American Academy of Implant Dentistry, and Educator for OCO Biomedical.

