Dr. Charles D. Schlesinger discusses a method for addressing the gap between an implant body and the residual socket
Today’s implant patient wishes to decrease overall treatment time as much as practitioners. Due to this fact, we are more frequently placing implants at the same time as an extraction is being done. By placing the implant at the same time as the extraction and not grafting first, there is a 4-6 month decrease in overall treatment time when compared to the later. Most roots differ in both length and three-dimensional form when compared to the typical cylindrical root-formed implant.
[userloggedin]
Grafting the gap
 Much has been written regarding how to manage the “gap” that results when an implant is placed in an extraction socket. When a dental implant is placed into a fresh extraction socket, the space between the implant periphery and surrounding bone is called the gap or jumping distance.1 Bone fill in the gap between the implant and the peripheral bone is important. The buccal aspect of an implant is of great concern, especially in the esthetic zone because the buccal bony plate is usually thin, and its resorption can result in soft tissue recession.2,3,4
Much has been written regarding how to manage the “gap” that results when an implant is placed in an extraction socket. When a dental implant is placed into a fresh extraction socket, the space between the implant periphery and surrounding bone is called the gap or jumping distance.1 Bone fill in the gap between the implant and the peripheral bone is important. The buccal aspect of an implant is of great concern, especially in the esthetic zone because the buccal bony plate is usually thin, and its resorption can result in soft tissue recession.2,3,4
The horizontal gap, if less than 2 mm, will likely fill in without any intervention by the practitioner. In fact, it has been shown that it is possible to have complete fill-in with a gap up to 4.2 mm, but this is not predictable.5
There are many materials on the market that are used to successfully fill this gap. Allograft, xenograft, and alloplastic materials all work, so it will come down to the clinical advantages and disadvantages of these materials along with their corresponding handling characteristics.
 OsteoGen® Strips
OsteoGen® Strips
The OsteoGen® Strips (Impladent Ltd.) are a combination of OsteoGen® non-ceramic bone graft crystals with bovine Achilles tendon collagen. The graft material is homogenously mixed with the collagen to create pliable sheets, which simplify placement and delivery of the graft while virtually eliminating the chance of particulate wash out. OsteoGen® Strips (Figure 1) were developed specifically to address clinical issues surrounding extraction/immediate placement of implants.
The graft inside these Plugs and Strips is OsteoGen®, a bioactive, non-ceramic (un-sintered) calcium-phosphate based graft with physicochemical properties similar to human bone mineral. The unique low temperature production process generates osteoconductive and resorbable low density crystals and crystal clusters with a unique calcium-to-phosphate ratio that is neither a beta-tricalcium phosphate nor a dense non-resorbable ceramic hydroxyapatite.6,7,8 OsteoGen® is highly hydrophilic, has been used as a particulate graft material since 1984, and has documented clinical success for use with implants in periodontal procedures,9,10 general osseous repair,11 and sinus lifts, including a decade-long longitudinal study of implant survival and success rates.12,13,14,15
The OsteoGen® Strips are radiolucent on the day of placement given their low-density properties; therefore, it is easy to follow the healing process radiographically as the site changes from radiolucent to radiopaque as the material is replaced by native bone over the course of 3-6 months.
Case study
A 63-year-old male with a non-contributory medical history presented with an endodontically treated upper right first bicuspid with recurrent decay (Figures 2 and 3). The tooth was evaluated clinically, and the pros and cons of trying to restore the tooth were discussed with the patient. Ultimately, the decision to extract the tooth and place an immediate implant was decided upon.
 After profound anesthesia was achieved with Septocaine® (Septodont USA), the tooth was atraumatically extracted (Figure 4) making sure not to fracture the bifurcated root. By making sure to not fracture the root, there were no potential issues that could have arisen if a more aggressive surgery was necessitated to remove a fractured root tip. The root dimensions were used to decide upon the final implant length and diameter (Figure 5). A purchase point was created with a high-speed bur to allow the pilot drill to be centered on the interseptal bone (Figure 6). A paralleling pin was placed to verify the position (Figure 7). The osteotomy was finalized utilizing a sequential osteotomy preparation (Figure 8). A 557 LS surgical carbide was used to perforate the lamina dura and produce adequate bleeding to establish a sufficient blood supply. OsteoGen® Strips were first saturated with the patient’s blood (Figure 9) before being placed in both buccally and lingually (Figures 10 and 11).
After profound anesthesia was achieved with Septocaine® (Septodont USA), the tooth was atraumatically extracted (Figure 4) making sure not to fracture the bifurcated root. By making sure to not fracture the root, there were no potential issues that could have arisen if a more aggressive surgery was necessitated to remove a fractured root tip. The root dimensions were used to decide upon the final implant length and diameter (Figure 5). A purchase point was created with a high-speed bur to allow the pilot drill to be centered on the interseptal bone (Figure 6). A paralleling pin was placed to verify the position (Figure 7). The osteotomy was finalized utilizing a sequential osteotomy preparation (Figure 8). A 557 LS surgical carbide was used to perforate the lamina dura and produce adequate bleeding to establish a sufficient blood supply. OsteoGen® Strips were first saturated with the patient’s blood (Figure 9) before being placed in both buccally and lingually (Figures 10 and 11).


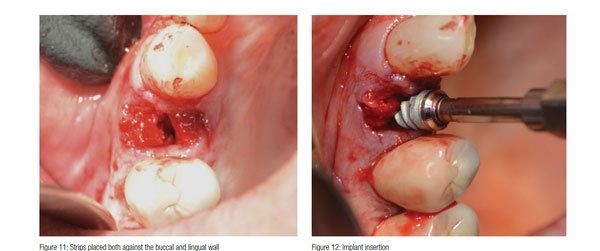 A 4.3 x 10 Hahn™ Tapered Implant (Glidewell Direct) was placed between the strips and into the prepared osteotomy (Figure 12). Excellent primary stability was attained with a seating torque value of 50N/cm and an ISQ of 68 utilizing an Osstell IDx, and the optimal position for restoration was achieved (Figure 13). According to the most recent literature published, an Osstell reading of 70 is desired for loading of an implant.16 The consistency of the strip no doubt contributed to the increased stability. Normally, this would be a good situation to immediately load this implant,17 but the decision to allow the graft material to mature first before loading in order to achieve the highest resistance to micro-motion was taken.
A 4.3 x 10 Hahn™ Tapered Implant (Glidewell Direct) was placed between the strips and into the prepared osteotomy (Figure 12). Excellent primary stability was attained with a seating torque value of 50N/cm and an ISQ of 68 utilizing an Osstell IDx, and the optimal position for restoration was achieved (Figure 13). According to the most recent literature published, an Osstell reading of 70 is desired for loading of an implant.16 The consistency of the strip no doubt contributed to the increased stability. Normally, this would be a good situation to immediately load this implant,17 but the decision to allow the graft material to mature first before loading in order to achieve the highest resistance to micro-motion was taken.
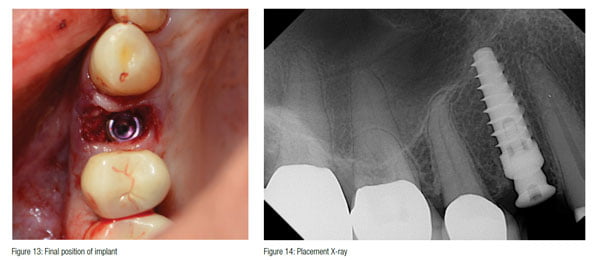

A 3 mm tall concave healing abutment was hand tightened (Figure 14), and the site was sutured with 4.0 pTfE sling suture (Figure 15).
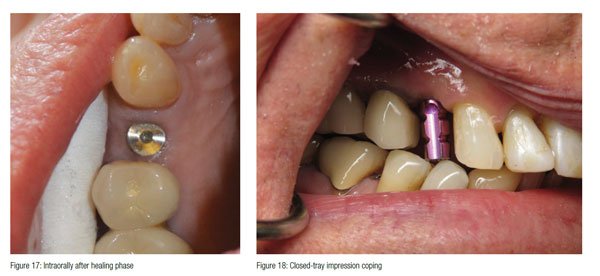
At 3 months, the implant was radiographed and demonstrated bone regeneration around the implant and crestal bone growth over the platform (Figure 16). The growth over the machined platform was made possible by the concave healing abutment and the initial sub-crestal platform position. The soft tissue was also fully healed, and sufficient keratinized tissue had matured around the healing abutment (Figure 17).
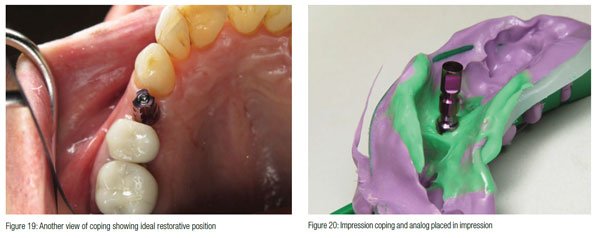
A closed tray impression coping was inserted (Figures 18 and 19), and a PVS impression was taken. An analog was placed on the impression coping after removal from the implant and inserted back into the impression (Figure 20). A shade was taken along with an opposing impression and bite registration before sending the case to the lab.
 One week later, the case returned from the lab, and the restorative dentist placed the abutment (Figure 21) and torqued it to 30 N/cm. Note the healthy keratinized tissue and gingival contour. The final full contour Zr crown was tried in and adjusted and finally cemented with resin-modified glass ionomer cement (Figures 22 and 23). The excess cement was removed, and the patient was dismissed.
One week later, the case returned from the lab, and the restorative dentist placed the abutment (Figure 21) and torqued it to 30 N/cm. Note the healthy keratinized tissue and gingival contour. The final full contour Zr crown was tried in and adjusted and finally cemented with resin-modified glass ionomer cement (Figures 22 and 23). The excess cement was removed, and the patient was dismissed.
Conclusion
 The immediate placement of a dental implant can be technically challenging. Attaining primary stability and preventing unwanted sequelae are priorities with this type of treatment. It is imperative to have a regenerative material that will not only fill the void between an implant and native bone, but also will not migrate from the site during healing.
The immediate placement of a dental implant can be technically challenging. Attaining primary stability and preventing unwanted sequelae are priorities with this type of treatment. It is imperative to have a regenerative material that will not only fill the void between an implant and native bone, but also will not migrate from the site during healing.
There are many ways to address the gap between an implant body and the residual socket; OsteoGen® Strips are an easy and cost-effective way to address these issues with outstanding results. The consistency of the product lends itself perfectly to the task by being stable, possibly enhancing initial stability, and providing a scaffold for new bone growth.
If immediately placing dental implants in extraction sites is part of your practice, do yourself a favor and take a look at this product.

- Botticelli D, Berglundh T, Buser D, et al. The jumping distance revisited: an experimental study in the dog. Clin Oral Implants Res. 2003;14(1):35-42.
- Januário AL, Duarte WR, Barriviera M, et al. Dimension of the facial bone wall in the anterior maxilla: a cone- beam computed tomography study. Clin Oral Implants Res. 2011;22(10):1168-1171.
- Katranji A, Misch K, Wang HL. Cortical bone thickness in dentate and edentulous human cadavers. J Periodontol. 2007;78(5):874-878.
- Chen ST, Darby IB, Reynolds EC. A prospective clinical study of non-submerged immediate implants: clinical outcomes and esthetic results. Clin Oral Implants Res. 2007; 18(5): 552-562.
- Rosenbach, DR. GAP management around immediate implants: a review of the literature and its application in clinical practice. Dentaltown. September 2014:44-49.
- Valen M, Ganz SD. A synthetic bioactive resorbable graft for predictable implant reconstruction: part one. J Oral Implantol. 2002;28(4):167-177.
- Artzi Z, Nemcovsky CE, Dayan D. Nonceramic hydroxyapatite bone derivative in sinus augmentation procedures: clinical and histomorphometric observations in 10 consecutive cases. Int J Periodontics Restorative Dent. 2003;23(4):381-389.
- Ricci JL, Blumenthal NC, Spivak JM, et al. Evaluation of a low temperature calcium phosphate particulate implant material: physicochemical properties and in vivo bone response. J Oral Maxillofac Surg. 1992;50(9):969-978.
- Epstein SR, Valen M. An alternative treatment for the periodontal infrabony defect: a synthetic bioactive resorbable composite graft. Dent Today. 2006;25(2):92-97.
- Corsair A. A clinical evaluation of resorbable hydroxylapatite for the repair of human intra-osseous defects. J Oral Implantol. 1990;16(2):125-128.
- Wagner JR. Clinical and histological case study using resorbable hydroxylapatite for the repair of osseous defects prior to endosseous implant surgery. J Oral Implantol. 1989;15:186-192.
- Fugazzotto PA, Vlassis JM. Long-term success of sinus augmentation using various surgical approaches and grafting materials. Int J Oral Maxillofac Implants. 1998;13(1):52-58.
- Vlassis JM, Hurzeler MB, Quinones CR. Sinus lift augmentation to facilitate placement of nonsubmerged implants: a clinical and histological report. Pract Periodontics Aesthet Dent. 1993;5(2):15-24.
- Whittaker JM, James RA, Lozada J, Cordova C, GaRey DJ. Histological response and clinical evaluation of heterograft and allograft materials in the elevation of the maxillary sinus for the preparation of endosteal dental implant sites. Simultaneous sinus elevation and root form implantation: an eight-month autopsy report. J Oral Implantol. 1989;15(2):141-144.
- Manso M, Wassal, T. A 10-year longitudinal study of 160 implants simultaneously installed in severely atrophic posterior maxillas grafted with autogenous bone and a synthetic bioactive resorbable graft. Implant Dent. 2010;19(4):351-360.
- Schlesinger C. Torque versus RFA at implant placement: a case study. Implant Practice US. 2016;9(4):14-20.
- Schlesinger C. Immediate placement of dental implants: a safe, a predictable treatment option. J Dent Sci. 2016;4(3).
Stay Relevant With Implant Practice US
Join our email list for CE courses and webinars, articles and mores

 Charles Schlesinger, DDS, FICOI, is a dental implant educator and clinician who has been lecturing internationally for the past 10 years. He graduated with honors from The Ohio State College of Dentistry in 1996. After graduation, he completed a General Practice residency at the VAMC San Diego and then went on to become the Chief Resident at the VAMC West Los Angeles. During his time in Los Angeles, he completed extensive training in oral surgery, implantology, and advanced restorative treatment. Once he completed his residency, Dr. Schlesinger maintained a thriving restorative and implant practice in San Diego, California for 14 years. In 2012, he relocated to Albuquerque, New Mexico to become the Director of Education and Clinical Affairs for OCO Biomedical. In 2013, he took over as Chief Operating Officer of OCO Biomedical along with his clinical responsibilities. In 2016, Dr. Schlesinger left OCO Biomedical and founded The CD Schlesinger Group, LLC, to help practitioners gain knowledge and experience with implants and became a Key Opinion Leader for The Hahn Implant System. Additionally, he continues to provide comprehensive implant care to patients in a private practice setting in Albuquerque, New Mexico.
Charles Schlesinger, DDS, FICOI, is a dental implant educator and clinician who has been lecturing internationally for the past 10 years. He graduated with honors from The Ohio State College of Dentistry in 1996. After graduation, he completed a General Practice residency at the VAMC San Diego and then went on to become the Chief Resident at the VAMC West Los Angeles. During his time in Los Angeles, he completed extensive training in oral surgery, implantology, and advanced restorative treatment. Once he completed his residency, Dr. Schlesinger maintained a thriving restorative and implant practice in San Diego, California for 14 years. In 2012, he relocated to Albuquerque, New Mexico to become the Director of Education and Clinical Affairs for OCO Biomedical. In 2013, he took over as Chief Operating Officer of OCO Biomedical along with his clinical responsibilities. In 2016, Dr. Schlesinger left OCO Biomedical and founded The CD Schlesinger Group, LLC, to help practitioners gain knowledge and experience with implants and became a Key Opinion Leader for The Hahn Implant System. Additionally, he continues to provide comprehensive implant care to patients in a private practice setting in Albuquerque, New Mexico.

