Announcing an Immediate Way for Surgeons to Deliver More Accurate
and Less Invasive Dental Implant Surgery to More Patients

Lansdale, PA, June 4, 2020 - X-Nav Technologies, LLC announced that it has received 510(k) Clearance from the U.S. Food & Drug Administration for X-Mark™, a new virtual patient registration technology that advances navigated dental implant surgery. X-Mark enhances the company’s industry-leading X-Guide® Dynamic 3D Navigation system to give dentists the ability to deliver more accurate dental implant procedures to more patients seeking new teeth.
X-Mark features breakthrough technology to facilitate virtual-based registration of the patient’s anatomy to a digital treatment plan. Developed to be fast and easy, the dentist will prepare for surgery by marking three virtual anatomy points on the patient’s 3D scan and then mark the same three live points on the patient at the time of surgery. The X-Guide system will use the X-Mark technology to quickly match the points together to enable live navigated surgery – with accuracy and ease.
X-Mark allows the dentist to provide all the benefits of less-invasive navigated surgery in more case presentations – from single tooth replacement to full mouth edentulous reconstruction.
The X-Guide system fits seamlessly into the dental office and works with all cone beam 3D scanners, including small field of view. It utilizes the surgeon’s digital plan to provide turn-by-turn guidance during surgery. X-Guide allows the surgeon to visualize precise 3D movements of the handpiece during osteotomy and implant delivery for more exact placement – like GPS for the dental drill. This delivers better functional and aesthetic results. With X-Guide navigation in the dental practice, more patients can benefit from same-day implant surgery, which is especially helpful as more patients want fast and smooth dental surgery.

Dentists can provide more immediate, more accurate, and less invasive dental implant options with X-Guide navigation.
“Over the past few years, the global dental industry has rapidly embraced the X-Guide dynamic 3D navigation technology because of its ability to deliver more accurate dental implant outcomes”, says Edward Marandola, President of X-Nav Technologies. “X-Mark virtual registration continues to advance and streamline the way surgeons place dental implants – while improving accuracy and facilitating better immediate restorative results. I am excited to launch X-Mark to expand the capabilities of the XGuide system so our customers can more easily offer navigation surgery to more patients.”
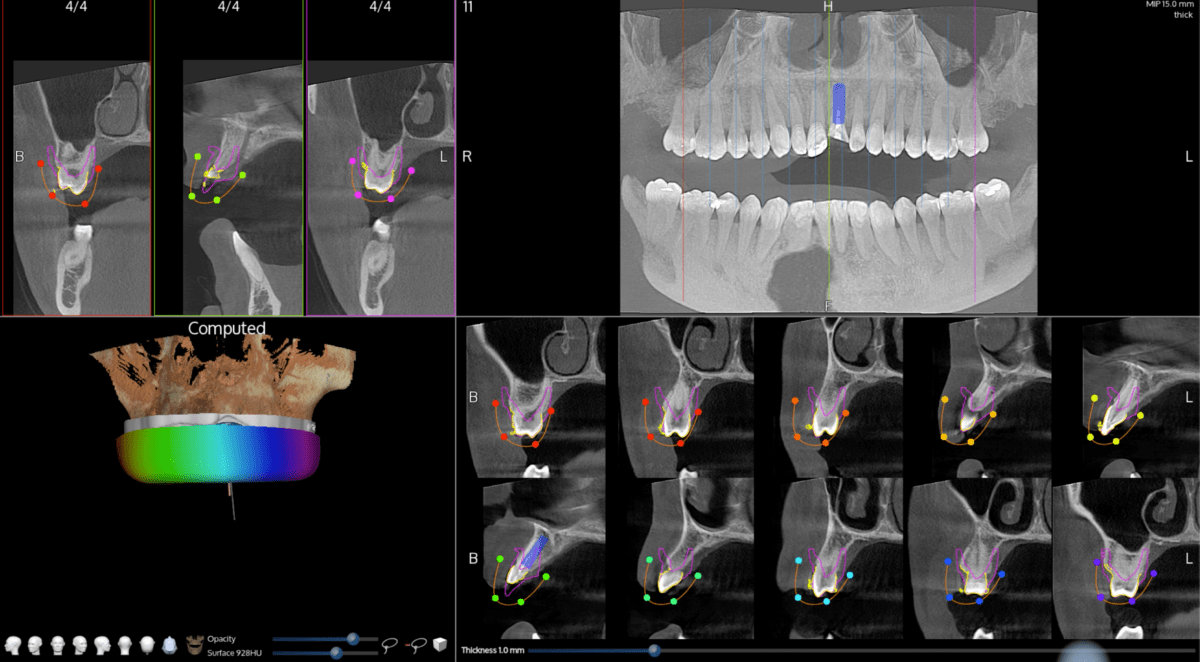
X-Mark for X-Guide is the industry’s first virtual patient registration process for dynamic dental navigation to receive 510(k) clearance from the U.S. Food and Drug Administration. X-Mark has also received CE Mark approval from the European Community, and Health Canada Medical Device License approval. The X-Guide Dynamic 3D Navigation system is used by dentists in over 35 countries and 20 languages – and still growing.

For more information about X-Guide Clinical Applications and Patient Information,
please visit www.X-NavTech.com.
About X-Nav Technologies, LLC
X-Nav Technologies is a medical device company that develops surgical products for the dental market that advance patient care while improving doctor productivity.
Stay Relevant With Implant Practice US
Join our email list for CE courses and webinars, articles and mores



