Robert Levine, DDS, and Peter Vitruk, PhD, discuss CO2 laser efficiency in coagulation and healing
Introduction
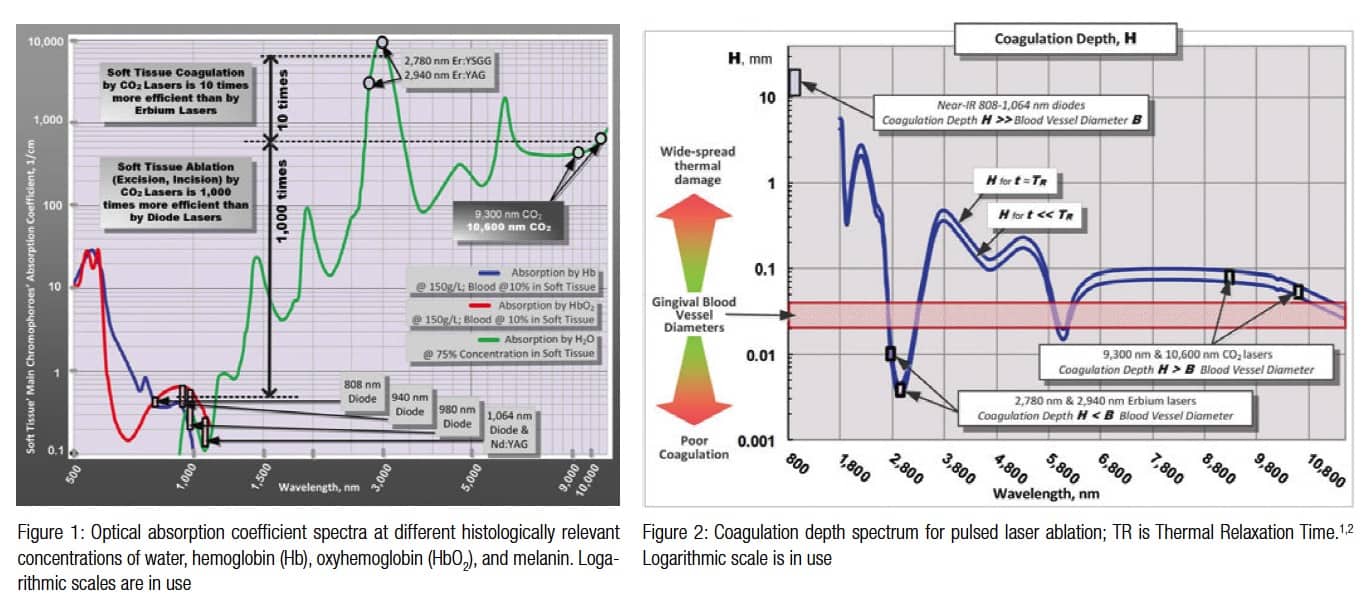
The key to successful applications of soft tissue lasers and their advantages over other surgical tools is their ability to accurately cut and efficiently coagulate the soft tissue at the same time. However, not all lasers are efficient at both cutting and coagulating. Some laser wavelengths (such as those of Erbium lasers) are great at cutting but are not as efficient at coagulating.1 Other wavelengths (such as those of diode lasers) are efficient coagulators but are poor scalpels.2 There are also lasers (such as the CO2 laser) that are efficient at both cutting and coagulating the soft tissue.1 The key to understanding how the laser light cuts and coagulates is through the wavelength-dependent nature of laser light’s absorption by the soft tissue, as presented in Figure 1 for the three wavelength groups of practical dental lasers that are on the market today:
- circa 1,000 nm (diodes and Nd:YAG laser)
- circa 3,000 nm (Erbium lasers)
- circa 10,000 nm (CO2 lasers)
Photo-thermal ablation and laser pulsing
The most efficient soft tissue laser ablation (and incision and excision) is a process of vaporization of intra- and extracellular water heated by the laser light within the irradiated soft tissue. Water vapors, rapidly steaming out of the intensely laser-heated soft tissue, carry with them cellular ashes and other byproducts of this fast boiling and vaporization process. Because of weak absorption (Figure 1) and strong scattering by the soft tissue,2 the near-infrared diode and Nd:YAG laser wavelengths circa 1,000 nm are highly inefficient and spatially in-accurate photo-thermal laser ablation tools.2 Because of very strong absorption by the soft tissue, mid-infrared Erbium (circa 3,000 nm) and infrared CO2 laser (circa 10,000 nm) wavelengths are highly efficient and spatially accurate laser ablation tools.1,2
The rate of how fast the irradiated tissue diffuses the heat away is defined by Thermal Relaxation Time TR, which equals approximately 1.5 msec for 75% water rich soft tissue irradiated by 10,600 nm CO2 laser. Practical implications of the Thermal Relaxation Time concept are simple and yet very powerful for appropriate application of laser energy. The most efficient heating of the irradiated tissue takes place when laser pulse energy is high and its duration is much shorter than TR. The most efficient cooling of the tissue adjacent to the ablated zone takes place if time duration between laser pulses is much greater than TR. Such laser pulsing is referred to as SuperPulse and is a must-have feature of any state-of-the-art soft tissue surgical CO2 laser that minimizes the depth of coagulation.1
Photo-thermal coagulation
Coagulation occurs as a denaturation of soft tissue proteins that occurs in 60º-100°C temperature range1 leading to a significant reduction in bleeding (and oozing of lymphatic liquids) on the margins of ablated tissue during laser ablation (and excision/incision) procedures. Since blood is contained within and transported through the blood vessels, the diameter of blood vessels B (estimated to range from 21 to 40 µm)3 is a highly important spatial parameter that influences the efficiency of photocoagulation process. Photo-thermal coagulation is also accompanied by hemostasis due to shrinkage of the walls of blood vessels (and lymphatic vessels) due to collagen shrinkage at increased temperatures. The coagulation depth H (for 60º-100ºC temperature range inside the ablation margins) was shown in Figure 1 to be proportional to the absorption depth A (an inverse of absorption coefficient presented in Figure 1) and is presented in Figure 2. The coagulation depth H relative to the blood vessel diameter B is an important measure of coagulation and hemostasis efficiency. For H<>B (diode laser wavelengths in Figure 2), optical absorption (Near-IR attenuation) and coagulation depths are significantly greater than blood vessel diameters; coagulation takes place over extended volumes.
For H ≥ B (CO2 laser wavelengths in Figure 2), coagulation extends just deep enough into a severed blood vessel to stop the bleeding. In another words, the CO2 laser’s excellent coagulation efficiency is due to the close match between the photo-thermal coagulation depth of approximately 50 µm,4 and oral soft tissue blood capillary diameters of approximately 20-40 µm.3 The key to the success of the soft tissue CO2 laser is its ability to cut and coagulate the soft tissue simultaneously.
CO2 laser oral soft tissue surgery
Figures 1 and 2 indicate that wavelengths circa 10,000 nm are >1,000 times superior to wavelengths circa 1,000 nm for soft tissue ablation and >10 times superior to wavelengths circa 3,000 nm for soft tissue coagulation and hemostasis. The 10,600nm CO2 laser is highly energy efficient at ablating the soft tissue photo-thermally with very low ablation threshold intensities due to extremely small volume of irradiated tissue because of extremely short absorption depth around 15 µm.1,2
Hemostasis and tissue healing
In comparison with conventional scalpel, the use of the CO2 laser in oral soft tissue surgeries offers several benefits. Among these benefits are cutting in a non-contact mode (which helps to avoid mechanical trauma to the tissue) and without bleeding or the need for sutures, precise tissue removal, and minimized postoperative pain, edema, and inflammation. The use of a CO2 laser on the oral soft tissue has no known contraindications or side effects.5 A substantial body of peer-reviewed literature describes the excellent hemostatic capacity of the CO2 laser as a useful instrument for oral surgery in patients with hemorrhagic disorders or undergoing antithrombotic therapy (Figures 3A-3B).5-8 Due to the hemostatic ability of the CO2 laser, intraoral surgical wounds often can be left to heal by secondary intention without placing sutures or dressing (as in Cases 3, 5, 7, and 8 described in this article).10 For example, the patient in Case 1 (Figures 3A-3C) wanted to have a vascular lesion (capillary hemangioma on the lip) removed. The risk of intraoperative hemorrhage was elevated by the fact that the patient had been taking blood thinning agents for his atrial fibrillation condition. In addition, the lesion was located in the area where successful cosmetic outcome was important; therefore, it was crucial to have good visualization of the operatory field. Efficient intra-operative hemostasis provided by the CO2 laser allows for more precise and accurate tissue removal due to the improved visibility of the surgical field (Figure 3B).9 In this case, the CO2 laser enabled the clinician to achieve the best results in a short period of time. No sutures were placed, and a thin layer of char was created to protect the surgical site. The recovery was uneventful. Figure 3C shows the completely healed surgical site at 1 month postoperatively.
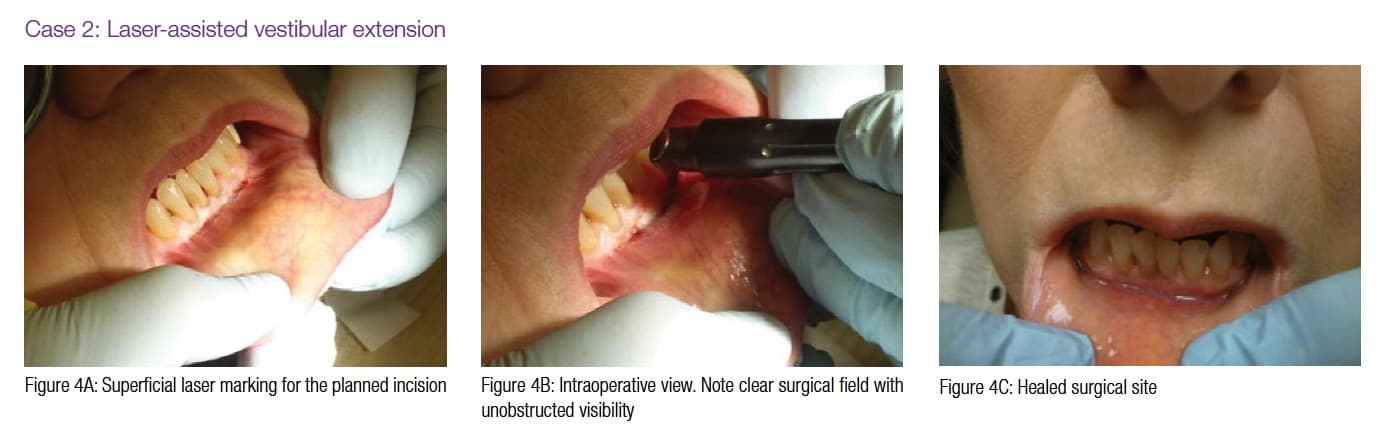
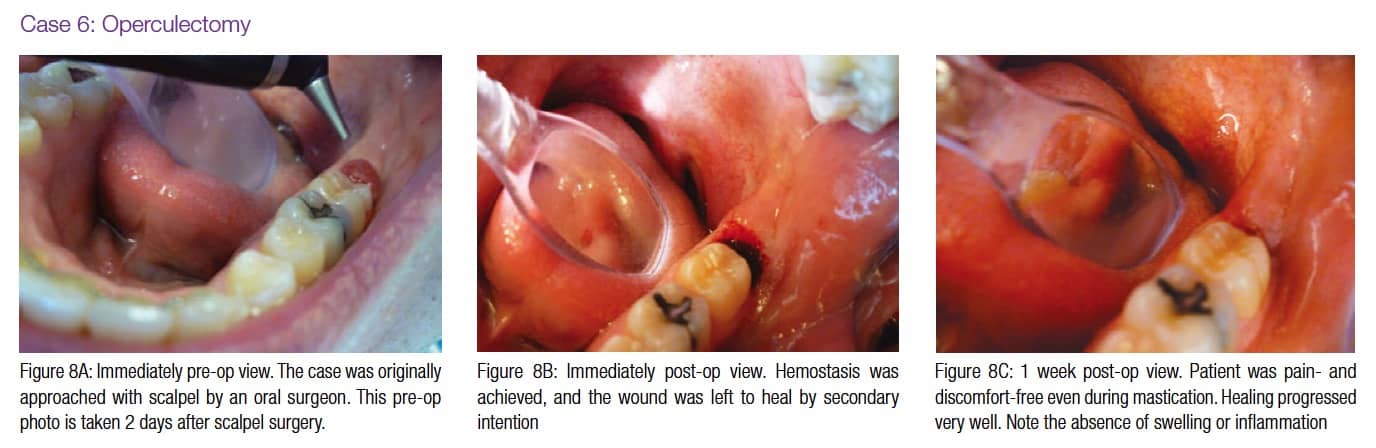
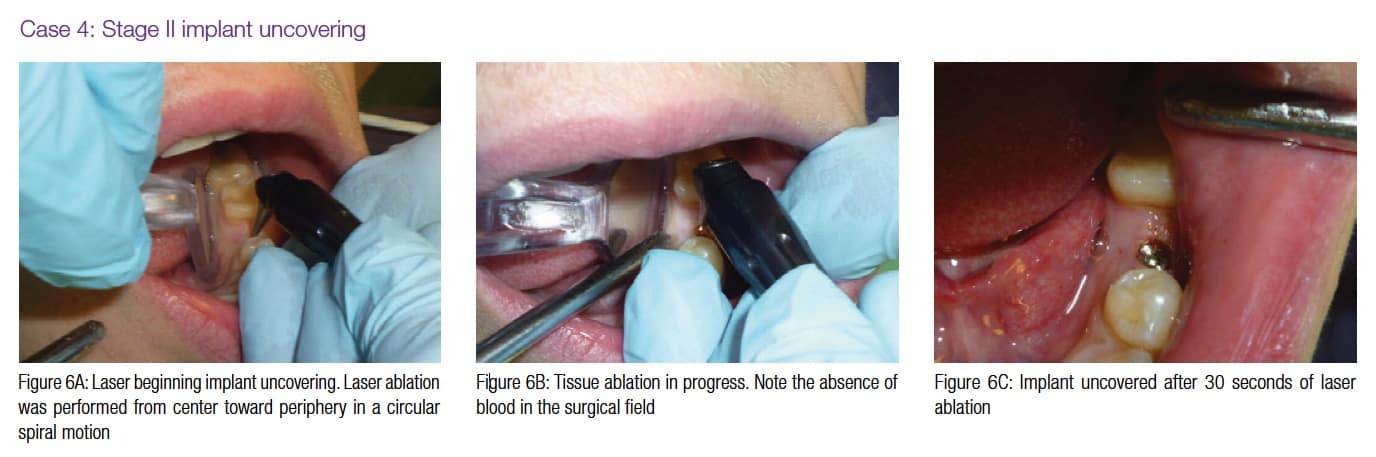
Figures 4A-4B, 5A-5B, 6A-6C, 7B-7C, and 8A demonstrate the enhanced hemostasis ensured by the LightScalpel CO2 laser. In Case 2, the patient had the vestibular extension procedure performed, and the hemorrhage was very well controlled, enabling the clinician with utmost precision. In Case 3, the bloodless excisional biopsy of fibroma was performed. Case 4 is a stage II implant uncovering procedure. This surgery requires both special accuracy and the well-controlled depth of laser energy penetration as well as enhanced hemostasis. No blood was present at any point. The implant was uncovered in approximately 30 seconds. In connection with implant uncovering, it should be mentioned that the recent study on the 10,600 nm CO2 laser removal of biofilm from titanium implant surfaces11 reported that this laser wavelength does not affect titanium or titanium oxide (most common implant materials) and is therefore safe to use around implants. Case 5 shows a LightScalpel CO2 laser lingual frenectomy in a 7-year-old patient. Surgery is performed in a highly vascularized area where bleeding was well controlled. Finally, Case 6 is the ablation of the inflamed operculum. Figure 8A demonstrates the immediately postoperative view with excellent hemostasis.
Similar to its effect on smaller blood vessels, the CO2 laser coagulates lymphatic vessels on the margins of the incision, which results in minimal postoperative swelling and edema, and ultimately, improved, less painful healing. Lymphatic vessels regenerate successively in approximately 8 to 10 days after capillary-vessel proliferation.12
Among the most prominent benefits of CO2 laser treatment are minimized wound contraction and scarring.10,13-16 Healing of CO2 laser-irradiated wounds is characterized by a more prominent fibroblastic proliferation, with young fibroblasts actively producing collagen. Compared to scalpel wounds, CO2 laser-induced wounds contain only a small number of myofibroblasts (cells responsible for wound contraction).16-18 According to Basu, et al.,19 and Tambuwala, et al.,20 healing of the CO2 laser-irradiated wounds involves the appearance of a fibroserous membrane 72 hours postoperatively. This membrane replaces the superficial necrotic layer of the laser-treated area. An epithelial covering of the wound starts to form from the periphery toward the center after two weeks and is thinner and parakeratotic in comparison with the epithelium that forms after scalpel resection. This could account for the excellent esthetic outcome of all CO2 laser treatment, with no fibrosis or scarring, and soft, pliable residual tissue.
Figures 3C (capillary hemangioma of the lip), 4C (mandibular vestibuloplasty), 5C (excisional biopsy of fibroma), and 8B-8C (operculectomy) depict beautiful healing of the CO2 laser surgical sites where the wound was left to heal by secondary intention. In the case of capillary hemangioma, the lack of scarring was important for esthetic reasons; in the case of vestibuloplasty, the absence of scarring was significant to preserve functionality; in the case of fibroma excision, smooth tissue ensured that the patient would not continue check biting and that the lesion would not reoccur.
Reduced wound contraction — combined with decreased lateral tissue damage, less traumatic surgery, more precise control of the depth of tissue damage, and excellent hemostatic ability — makes the CO2 laser a safe and efficient alternative to the conventional scalpel. Strauss, et al.,21 and Deppe, et al.,22 report that the healing process with CO2 laser surgery is faster and less painful than after cryosurgery or electrosurgery.
It should be mentioned, however, that the healing process for surgical laser wounds may be somewhat prolonged in comparison with surgical scalpel wounds. This delay in healing is believed to be caused by the sealing of blood vessels and lymphatics that subsequently requires neovascularization for healing. Typical intraoral healing takes 2 to 3 weeks for wounds that, if treated with a scalpel, normally would take 7 to 10 days.5, 22 At the same time, Lambrecht, et al.,12 reported a slightly shorter delay of just 3 to 10 days. The key to minimizing the healing time is through minimizing the thermal damage on the margins of laser incision/ablation (e.g., utilizing the SuperPulse CO2 laser settings).
Conclusion
The 10,600 nm CO2 laser is an efficient and spatially accurate photo-thermal ablation tool with excellent coagulation efficiency due to the close match between coagulation depth and oral soft tissue blood capillary diameters. The highly precise cutting with minimal collateral damage to the adjacent healthy tissue, good visibility of the operating field and, reportedly, relatively minimal postoperative pain make the CO2 laser an optimal surgical solution to the oral soft tissue surgery. In combination with the laser’s ability to cut in a non-contact mode, the risks of complications typically associated with outpatient treatment are reduced. The enhanced hemostatic capacity and improved healing with minimal to no scarring make the CO2 laser especially well suited for oral soft tissue surgery.
Acknowledgments
Authors greatly appreciate the support and contribution from Anna Glazkova, PhD, in preparing this material for publication.
This article is sponsored by LightScalpel, LLC.
(www.LightScalpel.com, 1-866-589-2722)
-
Vitruk P. Oral Soft Tissue Laser Ablative & Coagulative Efficiencies Spectra. Implant Practice US. 2014;7(6):22-27.
-
Vogel A, Venugopalan V. Mechanisms of pulsed laser ablation of biological tissues. Chem Rev. 2003;103(2):577-644.
-
Yoshida S, Noguchi K, Imura K, Miwa Y, Sunohara M, Sato I. A morphological study of the blood vessels associated with periodontal probing depth in human gingival tissue. Okajimas Folia Anat Jpn. 2011;88(3):103-109.
-
Convissar RA. Principles and Practice of Laser Dentistry. St. Louis, MO: Mosby Elsevier 2011.
-
Gama SK, de Araújo TM, Pinheiro AL. Benefits of the use of the CO2 laser in orthodontics. Lasers Med Sci. 2008;23(4):459-465.
-
Coleton S. Lasers in surgical periodontics and oral medicine. Dent Clin North Am. 2004;48(4):937-962.
-
Monteiro LS, Mouzinho J, Azevedo A, Câmara MI, Martins MA, La Fuente JM. Treatment of epulis fissuratum with carbon dioxide laser in a patient with antithrombotic medication. Braz Dent J. 2012;23(11):77-81.
-
Tuncer I, Ozçakir-Tomruk C, Sencift K, Cöloğlu S. Comparison of conventional surgery and CO2 laser on intraoral soft tissue pathologies and evaluation of the collateral thermal damage. Photomed Laser Surg. 2010;28(1):75-79.
-
Kotlow LA. Lasers in pediatric dentistry. Dent Clin North Am. 2004;48(4):889-922.
-
Zaffe D, Vitale MC, Martignone A, Scarpelli F, Botticelli AR. Morphological, histochemical, and immunocytochemical study of CO2 and Er:YAG laser effect on oral soft tissues. Photomed Laser Surg. 2004;22(3):185-189.
-
Cobb CM, Vitruk P. Microbial Decontamination of Three Different Implant Surfaces Using a SuperPulse CO2 (10,600 nm) Laser: An In Vitro Study. Presented at the Academy of Laser Dentistry Meeting; Feb. 5-7, 2015; Palm Springs, CA.
-
Lambrecht JT, Stübinger S, Hodel Y. Treatment of intraoral hemangiomas with the CO2 laser. J Oral Laser Appl. 2004;4:89-96.
-
Zeinoun T, Nammour S, Dourov N, Aftimos G, Luomanen M. Myofibroblasts in healing laser excision wounds. Lasers Surg Med. 2001;28(1):74-79.
-
Mason C, Hopper C. The use of CO2 laser in the treatment of gingival fibromatosis: a case report. Int J Paediatr Dent. 1994;4(2):105-109.
-
Wang X, Ishizaki NT, Matsumoto K. Healing process of skin after CO2 laser ablation at low radiance: a comparison of continuous-wave and pulsed mode. Photomed Laser Surg. 2005;23(1):20-26.
-
Grbavac RA, Veeck EB, Bernard JP, Ramalho LM, Pinheiro AL. Effects of laser therapy in CO2 laser wounds in rats. Photomed Laser Surg. 2006;24(3):389-396.
-
de Freitas AC, Pinheiro AL, de Oliveira MG, Ramalho LM. Assessment of the behavior of myofibroblasts on scalpel and CO2 laser wounds: an immunohistochemical study in rats. J Clin Laser Med Surg. 2002;20(4):221-225.
-
Fisher SE, Frame JW, Browne RM, Tranter RM. A comparative histological study of wound healing following CO2 laser and conventional surgical excision of canine buccal mucosa. Arch. Oral Biol. 1983;28(4):287-291.
-
Basu MK, Frame JW, Rhys Evans PH. Wound healing following partial glossectomy using the CO2 laser, diathermy and scalpel: a histological study in rats. J Laryngol Otol. 1988;102(4):322-327.
-
Tambuwala A, Sangle A, Khan A, Sayed A. Excision of oral leukoplakia by CO2 lasers versus traditional scalpel: A comparative study. J Maxillofac Oral Surg. 2014;13(3):320-327.
-
Strauss RA, Fallon SD. Lasers in contemporary oral and maxillofacial surgery. Dent Clin North Am. 2004;48(4):861-888.
-
Deppe H, Horch HH. Current status of laser applications in oral and cranio-maxillofacial surgery. Med Laser Appl. 2007;22(1):39-42.
Stay Relevant With Implant Practice US
Join our email list for CE courses and webinars, articles and mores