Charles M. Cobb, DDS, MS, PhD, and Peter Vitruk, PhD, quantify implant surface decontamination parameters
Abstract
Background: As dental implants become a routine part of dental practice, so too will the prevalence of peri-implant diseases. Inherent to the treatment of peri-implant disease is the removal of microbial biofilms from the implant surface. Currently, there is no standardized protocol for application of any treatment modality directed at implant surface de-contamination.
[userloggedin]
In this in vitro study, we report on the effectiveness of a super-pulsed CO2 laser (10.6 μm wavelength), delivering an average fluence of 6.3 to 113 J/cm2, to remove biofilm from three different types of implant surface topographies.
Methods: Sixty-six implants representing three distinctly different surface topographies were used to prepare 132 specimens yielding 44 of each surface type. A 48-hour mixed species biofilm was established on the surface of each specimen. Controls consisted of untreated specimens while treated specimens were irradiated at eight different laser energy density (or fluence) levels ranging from 6.3 to 113 J/cm2. Outcomes were measured by SEM examination and CFU counts of residual microbes following treatment at each fluence.
Results: Biofilms ranged in thickness from 5 to 15 μm. An average fluence of 19 J/cm2 was sufficient to achieve 100% ablation of the biofilm on hydrophilic sandblasted and acid-etched surface specimens (SA). However, to achieve 100% ablation of biofilm on HA and highly crystalline, phosphate enriched titanium oxide (PTO) surfaced implants required an average fluence of 38 J/cm2.
Conclusions: In this in vitro model, a 10.6 μm wavelength super-pulse CO2 laser using parameters that produce an average fluence of 38 J/cm2 will achieve 100% ablation of biofilms grown on implant specimens of varying topographies.
Introduction
As the placement of dental implants increases and becomes a routine part of dental practice so too will the prevalence rate of peri-implant diseases.1 Some reports suggest the radiographic evidence of peri-implant bone loss may reach 30% to 35% while others report a lower rate, ranging from 10% to 20%.2-7 The treatment of peri-implantitis, at some point, requires removal of all microbial biofilms from the exposed implant surface that, in turn, allows for optimal healing of host bone, osseous regeneration techniques, and overlying soft tissues.
Various methods of implant surface decontamination have been suggested, including use of tetracycline or sterile saline-soaked cotton pellets,8-11 air-polishing with sodium bicarbonate,12 diode,13 Nd:YAG,13 Er:YAG,14 Er,Cr:YSGG,15 and CO2 lasers.16 In addition, various adjunctive combinations with flap surgery and/or regenerative therapies have also been investigated.8-10, 17,18 None of the prescribed methods of implant surface decontamination has been shown to be superior in the management of peri-implantitis. Part of the problem relates to the fact that there are no standardized protocols for any treatment modality directed at implant surface decontamination, including use of the CO2 laser. Thus, the purpose of this in vitro project was to establish the minimum fluence at which complete microbial decontamination can be achieved using a super-pulsed CO2 laser on each of three different, but common, implant surface topographies.
Materials and methods
Implants
Three implants types were selected that collectively represented a range of topography and surface roughness (Figure 1). The implants’ surfaces were as follows:
- hydroxyapatite (HA) plasma-sprayed (Steri-Oss® HA Coated Surface, Steri-Oss Inc.)
- a highly crystalline, phosphate enriched titanium oxide surface produced by spark anodization (PTO) (NobelReplace™ Groovy, Nobel Biocare® USA)
- hydrophilic sandblasted and acid-etched surface (SA) (Straumann® SLActive® Surface, Straumann USA, LLC)
From a total of 66 intact implants (22 of each surface type), two specimens measuring 1.0 mm (thickness) x 1.6 mm x 4.0 mm were cut from each 4.8 mm or 5.0 mm x 10 mm implant using a low-speed diamond saw (Buhler IsoMet® saw, Buhler). Specimens were cleaned of residual debris from the cutting process by sonication for 1 minute and rinsing twice, using sterile water. All implant specimens were sterilized by UV light exposure for 60 minutes19 and then stored in sterile 48-well flat-bottomed cell culture dishes (Falcon® Cultureware, BD Biosciences) until inoculated and incubated with broth-containing bacteria.
A total of 44 specimens of each implant surface type were divided as follows:
- one untreated negative control (no biofilm for SEM), one untreated positive control (with biofilm for SEM), and one specimen from each of the eight laser treatment groups, all examined by SEM
- two untreated positive controls from each surface type to establish a baseline CFU count
- four treated specimens of each surface type from each of the eight different laser treatment groups used for CFU counts.
Biofilm
Four supragingival plaque samples were obtained from each of five private practice periodontal patients exhibiting moderate and/or severe chronic periodontitis (i.e., ≥ 5 mm probing depth with bleeding on probing and radiographic evidence of interproximal bone loss). Samples were obtained during scaling and root planing appointments using a McCall 13S/14S curette. The technique and purpose of the plaque sampling was explained in detail to the volunteer patients.
All patients signed an informed consent according to the Helsinki Declaration.20
Plaque samples were pooled in 100 ml of broth media (Terrific Broth, Invitrogen®/Life Technologies) and homogenized by vortex agitation for 1 minute. The pooled plaque sample was then incubated in a 150 ml roller bottle using a standard roller bottle apparatus under microaerophilic conditions at 37ºC for 48 hours. Following incubation, the 100 ml sample was agitated by vortex for 1 minute and then divided into four equal aliquots of 25 ml each. Fresh broth media was then added to each 25 ml aliquots, adjusting each to an absorption density of 1.0 (λ 630 nm) yielding four tubes of equal bacterial densities that were again agitated by vortex for 30 seconds. The resulting tubes of mixed bacteria were used as the stock sample for inoculation of the implant specimens. The inoculum contained approximately 106 microbes per ml.
Immediately prior to inoculating implant specimens with the bacteria-ladened broth, they were immersed for 2 hours in fetal bovine serum (Gibco® Fetal Bovine Serum, Life Technologies), which was diluted 1:1 in 25% sterile physiological saline. Preconditioning with fetal bovine serum served as a surrogate for GCF and salivary proteins that implants would normally acquire in situ. Using a 48-well sterile culture dish, three implant specimens of each surface type were placed in a single well and covered with 1.0 ml of stock solution (i.e., broth media) containing bacteria. The “inoculated” implants were then incubated on a shaker table (low setting for gentle agitation) at 37ºC in a microaerophilic environment for 48 hours. The resulting biofilm attached to the implant specimens ranged in thickness from 5 to 15 μm.
Laser
The laser used in this project was a Super-Pulsed CO2 laser (10,600 nm) (Luxar NovaPulse LX-20SP CO2 laser, LightScalpel, LLC). The laser handpiece LightScalpel PN LS9002-07 was fitted with a 0.8 mm spot size diameter ceramic delivery tip LightScalpel PN LS9005-01 (86% of beam energy contained within 0.8 mm diameter, 100% of energy contained within 1 mm diameter). The eight different fluence levels were determined by using the parameters listed in Table 1 (laser settings “A” through “H”).
All laser settings were for SuperPulse mode (peak power > 50 W, pulse widths under 800 μsec, and pulse repetition rates of 150Hz. SuperPulse modes were further gated (“Repeat” gate settings, Table 1 and Figure 2A) with longer pulses (≥ 10 msec) at lower frequencies (≤ 30 Hz).
The pulse-width and pulse-rate settings of SuperPulse mode allow for thermal confinement of the laser energy deposited into 5-15 μm thick water-rich biofilm as will be discussed below.
A previous study21 determined that when manually using a CO2 laser to “paint” a flat surface, the typical rate of irradiation exposure is 4 mm/second. Thus, as all specimens measured 4 mm in length and 1.6 mm in width, each of two non-overlapping sweeps of the laser beam was of 1-second duration.
The laser handpiece was stabilized at a fixed 2 mm distance over the target surface by use of an adjustable-angle clamp attached to an L-shaped base support stand. Implant specimens were placed on a glass microscope slide that, in turn, was positioned on a variable speed motorized microscope stage, allowing passage of the specimen under the laser beam at the prescribed rate of 4 mm/second. This arrangement also allowed for minimal overlapping of exposed surfaces during the second pass required to cover the 1.6 mm width using the 0.8 mm diameter beam delivery tip (Figure 2B).
Following laser treatment of the implant specimen surface, the edges of the specimen were irradiated using a continuous beam and 6 Watts. This was to ensure that bacteria attached to the edges were ablated. Thus, only residual bacteria from the treated surface, if any, contributed to CFU counts.
Immediately following laser treatment, one specimen of each surface type from each treatment group was selected for SEM evaluation and immersed in fixative. The remaining three specimens were immersed in sterile broth media and processed for CFU counts. A summary schematic of the experimental protocol is presented in Figure 2C.
Colony-forming units
All laser treated implant specimens, including the eight untreated positive control specimens (with biofilm), were processed in a sterile laboratory hood in the following manner:
- One implant specimen from each laser treatment group was randomly selected for SEM evaluation.
- The remaining implant specimens were each immersed in 10 ml of fresh broth media and vigorously agitated by vortex for 2 minutes followed by 1 minute of sonication.
- Serial dilutions were made from the supernatant (1:10,000 and 1:100,000).
Aliquots of 0.1 ml from each of the two dilutions were plated manually in triplicate using a spiral platter onto non-selective blood agar plates (Sigma-Aldrich) supplemented with hemin (5 mg/ml), menadione (1 mg/ml), and 5% sterile sheep blood. Plates were incubated under microaerophilic conditions at 37ºC for 72 hours.
Following incubation, the total number of CFU/ml was determined for each plate using an electronic colony counter system (Scienceware® Electronic Colony Counter System, Bel-Art Products). All counts were converted assuming a dilution of 1:100,000 and recorded as the average number of CFUs/ml. There was no attempt to sub-culture or identify recovered microbes other than by SEM morphotype.
Scanning Electron Microscopy
Specimens dedicated to SEM evaluation were immersed in ice-cold fixation consisting of 2.5% gluteraldehyde in 0.1 M cacodylate buffer at pH 7.4 for 2 hours. Following fixation, specimens were rinsed 3 times in 0.1 M cacodylate buffer (pH 7.4) for 5 minutes per rinse. Following the buffer rinse, specimens were dehydrated in a series of graded ethanol solutions (20% to 100%) at 5-minute intervals, followed by immersion in hexamethyldisilazane for 30 minutes. Each specimen was then affixed to an aluminum stub and stored in a desiccator overnight, followed by sputter-coating with approximately 20 nm of gold-palladium. Specimens were examined in a XL30 ESEM-FEG scanning electron microscope (FEI Corp., North America NanoPort) at various magnifications ranging from x80 up to x4,000. Magnifications of 2,000x and 4,000x were used to identify residual microbes by morphotype, i.e., coccus, short, medium, and long rods, fusiform, spirochete-like, and curved rods.
Statistical analysis
Regardless of implant surface topography, total decontamination of the implant surface is the clinical goal when treating peri-implant disease. Thus, the CFU data were log transformed to obtain only the main effect for each level of laser treatment. Descriptive statistics are presented for raw data and log-transformed CFUs. The measured outcome of laser treatment is the percentage reduction in CFU counts, assuming the baseline count of untreated implant specimens to represent 100%.
Results
The 48-hour biofilm that developed on the implant specimens ranged in thickness from 5 to 15 μm and was dominated by microbial morphotypes that were typically associated with a supragingival biofilms of natural teeth, i.e., cocci, short and medium length rods (Figure 3A). The morphologic character and thickness of the biofilm was consistent for all implant specimens regardless of the surface topography.
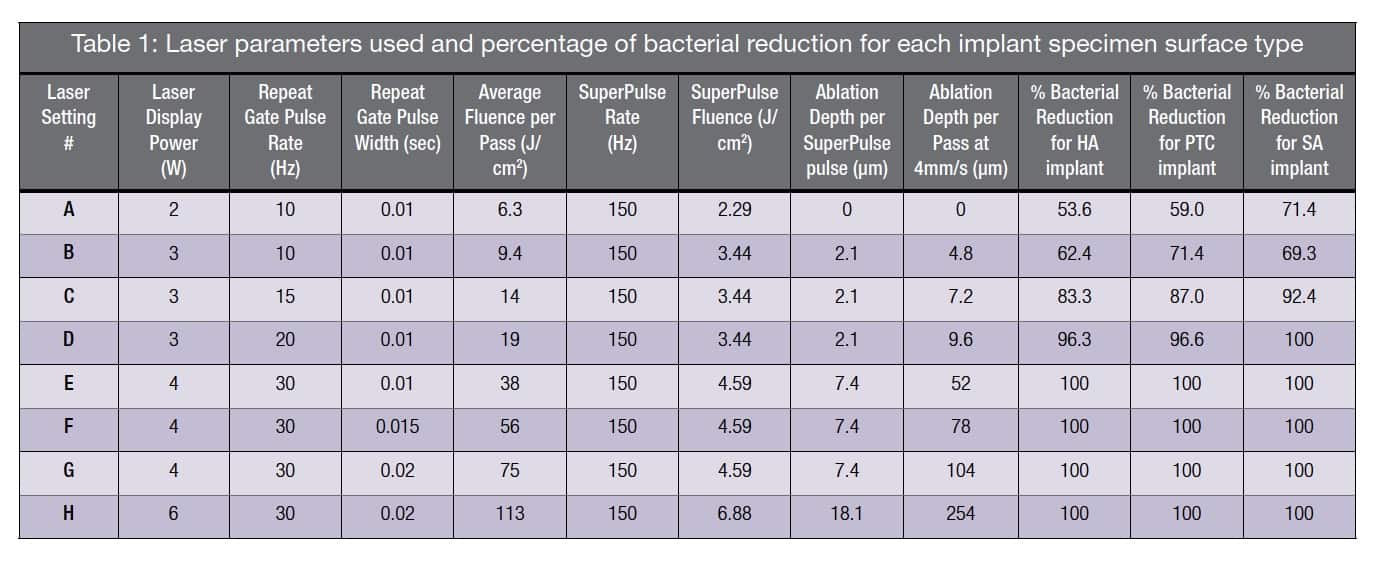 Table 1 summarizes the laser setting parameters and the results of the bacterial reduction, as well as the calculated depth of the laser ablation. It should be noted that 100% bacterial reduction was achieved at a fluence of 19 J/cm2 (laser setting “D”) per passage of the laser beam for the SA implant surface and for both the HA and PTO implant surfaces the bacterial reduction was 96.3% and 96.6%, respectively. At a fluence of 38 J/cm2 (laser setting “E”) or greater per pass, 100% of the biofilm was ablated and 100% bacterial reduction was measured regardless of the type of implant surface.
Table 1 summarizes the laser setting parameters and the results of the bacterial reduction, as well as the calculated depth of the laser ablation. It should be noted that 100% bacterial reduction was achieved at a fluence of 19 J/cm2 (laser setting “D”) per passage of the laser beam for the SA implant surface and for both the HA and PTO implant surfaces the bacterial reduction was 96.3% and 96.6%, respectively. At a fluence of 38 J/cm2 (laser setting “E”) or greater per pass, 100% of the biofilm was ablated and 100% bacterial reduction was measured regardless of the type of implant surface.
 Use of per pass beam fluence of 6.3. 9.4, and 14 J/cm2 (laser settings “A-C”) achieved incomplete biofilm ablation and bacterial reduction, ranging from a low of 53.6% up to 92.4% with little variation between implant surface types (Table 1). SEM examination of specimens treated with a fluence of
Use of per pass beam fluence of 6.3. 9.4, and 14 J/cm2 (laser settings “A-C”) achieved incomplete biofilm ablation and bacterial reduction, ranging from a low of 53.6% up to 92.4% with little variation between implant surface types (Table 1). SEM examination of specimens treated with a fluence of
≤ 14 J/cm2 revealed the presence of residual microbes (incomplete biofilm ablation) within surface depressions (Figures 3B and 3C). At a beam fluence of 38 J/cm2 (laser setting “E”), it was noted that the HA surface exhibited evidence of melting (Figure 3D), whereas the PTO and SA surfaces showed no surface alterations at any of the higher fluence exposures, i.e., 38, 56, 75, and 113 J/cm2.
Discussion
Efficient bacterial reduction can be achieved by ablating the bacterial biofilm off the implant surface without heating nor damaging the implant if:
- Laser energy is efficiently deposited into the biofilm.
- Laser-generated heat inside the biofilm is confined to the biofilm and is not thermally conducted away into the body of the implant (which acts as a highly efficient heat sink).
- Laser-generated heat inside the biofilm is sufficient for vaporizing the biofilm.
 The first condition (efficient laser energy deposition into the biofilm) is met when laser absorption depth is comparable to the biofilm’s thickness. Figure 4 presents absorption depth and thermal relaxation time spectra22 for a biofilm assumed to have a water content of 85%.23 The absorption depth of a 10.6 µm wavelength CO2 laser wavelength is about 14 µm, which is well within the range of the biofilm thickness observed in this study, i.e., range of 5 to 15 µm.
The first condition (efficient laser energy deposition into the biofilm) is met when laser absorption depth is comparable to the biofilm’s thickness. Figure 4 presents absorption depth and thermal relaxation time spectra22 for a biofilm assumed to have a water content of 85%.23 The absorption depth of a 10.6 µm wavelength CO2 laser wavelength is about 14 µm, which is well within the range of the biofilm thickness observed in this study, i.e., range of 5 to 15 µm.
The second condition of thermal confinement of laser energy within the irradiated biofilm is met when the laser pulse duration is shorter than Thermal Relaxation Time (TRT), also presented in Figure 4. TRT defines the rate of how fast the irradiated tissue diffuses the heat away as defined through the thermal diffusion time: TRT = A2/K,24,25 where A is optical absorption depth discussed above. The physics behind thermal diffusivity process is similar to diffusion and Brownian motion first described by Einstein in 1905.26 Co-efficient K is the tissue’s thermal diffusivity; K = λ /(ϱ C) ≈ 0.155 (+/-0.007) mm2/sec (derived from heat conductivity λ ≈ 6.2-6.8 mW/cm ºC; specific heat capacity C ≈ 4.2 J/g ºC, and density ϱ ≈ 1 g/cm3 for liquid water for temperatures in 37ºC-100ºC range).27 For 10 µm thick 80%-90% water-rich biofilm at 10,600 nm wavelength, the TRT is approximately 700 µsec. Therefore, a SuperPulsed CO2 laser with pulses under 800 µsec are highly efficient at confining the heat generated by the laser pulse within the biofilm thickness during the laser pulse.
The third condition of efficient biofilm vaporization (or ablation) is met when laser fluence during the SuperPulse pulse exceeds laser ablation threshold Eth25,28 presented in Figure 4. The more efficiently the laser energy is absorbed (Erbium and CO2 lasers), the lower is the ablation threshold. The less efficient the laser energy is absorbed (diode and Nd:YAG lasers), the higher is the ablation threshold. The ablation threshold at the 10.6 µm CO2 laser’s wavelength for a biofilm with an assumed 85% water content equals approximately 3 J/cm2. Laser settings “B-H” from Table 1 with fluence over 3 J/cm2 fit the ablative requirements.
During each SuperPulse pulse, the ablation depth δ is given by the formula δ = A (E – Eth) / Eth for the steady state ablation conditions,25 where A is the absorption depth and Eth is the ablation threshold fluence, and E is the fluence during the SuperPulse pulse. Ablative laser settings “B-D” in Table 1 (with fluence over 3 J/cm2) allow for an ablation depth of approximately 2-18 µm per pulse; multiple pulses stacked on top of each other allow for deeper ablation depths (proportional to number of pulses).
Bacterial reduction depends on how much of the bacterial biofilm is ablated as illustrated in Figure 5. Bacterial reduction of 100% can be achieved only through complete laser ablation of the biofilm; the sub-ablative laser setting “A” with 2.29 J/cm2 is seen as the least efficient.
As also observed in this study, plasma-sprayed hydroxyapatite HA implant surface is susceptible to melting and heat-induced cracking at an average fluence greater than 19 J/cm2 and certainly at the 38 J/cm2 as noted in this study. This can be explained through high absorption of the 10,600 nm light by the hydroxyapatite29 exposed after the bacterial biofilm was ablated. In contrast, the PTO and SA surfaces did not exhibit adverse surface interactions even at higher fluences up to 113 J/cm2, which can be explained by high reflectivity of titanium (> 90%) at 10,600 nm.30
Conclusion
An average fluence of 19 J/cm2 delivered by a SuperPulse 10.6 μm wavelength CO2 laser is sufficient to achieve a 100% ablation of an in vitro biofilm of approximately 10 μm thickness grown on implant specimens with a moderately rough surface topography. A similar reduction on HA or highly crystalline, phosphate enriched titanium oxide surfaces required an average fluence of 38 J/cm2. The HA surface implant specimens also exhibited surface melting and heat crazing at an average fluence of 38 J/cm2. The SuperPulse 10.6 μm wavelength CO2 laser may provide a predictable method of surface decontamination in the treatment of peri-implantitis.
[/userloggedin]
[userloggedout][/userloggedout]
- Da Silva JD, Kazimiroff J, Papas A, Curro FA, Thompson VP, Vena DA, Wu H, Collie D, Craig RG. Outcomes of implants and restorations placed in general dental practices: A retrospective study by the Practitioners Engaged in Applied Research and Learning (PEARL) Network. J Am
Dent Assoc. 2014;145(7):704-713. - Roccuzzo M, Bonino F, Aglietta M, Dalmasso P. Tenyear results of a three arms prospective cohort study on implants in periodontally compromised patients. Part 2: Clinical result. Clin Oral Implants Res. 2012;23(4):389-395.
- Renvert S, Lindahl C, Rutger-Persson G. The incidence of peri-implantitis for two different implant systems over a period of thirteen years. J Clin Periodontol. 2012;39(12):1191-1197.
- Fardal O, Grytten J. A comparison of teeth and implants during maintenance therapy in terms of the number of disease-free years and costs – an in vivo internal control study. J Clin Periodontol. 2013;40(6):645-651.
- Gruica B, Wang HY, Lang NP, Buser D. Impact of IL-1 genotype and smoking status on the prognosis of osseointegrated implants. Clin Oral Implants Res. 2004;15(4):393-400.
- Koldsland OC, Scheie AA, Aass AM. Prevalence of periimplantitis related to severity of the disease with different degrees of bone loss. J Periodontol. 2010;81(2):231-238.
- Mombelli A, Müller N, Cionca N. Epidemiology of periimplantitis. Clin Oral Implants Res. 2012;23(Suppl 6):67-76.
- Schmidt EC, Papadimitriou DEV, Caton JG. Surgical management of peri-implantitis: A clinical case report. Clin Adv Periodontics. 2014;4(1):31-37.
- Aljateeli M, Fu JH, Wang HL. Managing peri-implant bone loss: current understanding. Clin Implant Dent Related Res. 2012;14(Suppl 1):e109-e118.
- Froum SJ, Froum SH. Rosen PS. Successful management of peri-implantitis with a regenerative approach: A consecutive series of 51 treated implants with 3- to 7.5-year followup. Int J Periodontics Restorative Dent. 2012;32(1):11-20.
- Heitz-Mayfield LJ, Salvi GE, Mombelli A, Faddy M, Lang NP. Anti-infective surgical therapy of peri-implantitis. A 12-month prospective clinical study. Clin Oral Implants Res. 2012;23(2):205-210.
- Vouros JD, Papadopoulos C, Menexes G, Konstantinidis A. Laser and air-abrasive therapies in the nonsurgical treatment of peri-implantitis: A systemic review. J Implant Adv Clin Dent 2014;6:21-36.
- Goncalves F, Zanetti AL, Zanetti RV, Martelli FS, Avila- Campos MJ, Tomazinho LF, Granjeiro JM. Effectiveness of 980-mm diode and 1064-nm extra-long-pulse neodymiumdoped yttrium aluminum garnet lasers in implant disinfection. Photomed Laser Surg. 2010; 28(2):273-280.
- Yamamoto A, Tanabe T. Treatment of peri-implantitis around TiUnite-surface implants using Er:YAG laser microexplosions. Int J Periodontics Restorative Dent. 2013;33(1):21-30.
- Schwarz, F, Nuesry E. Bieling K. Herten M., Becker J. Influence of an erbium, chromium- doped yttrium, scandium, gallium, and garnet (Er,Cr:YSGG) laser on the reestablishment of the biocompatibility of contaminated titanium implant surfaces. J Periodontol. 2006;77(11): 1820-1827.
- Romanos GE, Nentwig GH . Regenerative therapy of deep peri-implant infrabony defects after CO2 laser implant surface decontamination. Int J Periodontics Restorative Dent. 2008;28(3):245-255.
- Schwarz F, Sahm N, Iglhaut G, Becker J. Impact of the method of surface debridement and decontamination on the clinical outcome following combined surgical therapy of peri- implantitis: a randomized controlled clinical study. J Clin Periodontol. 2011;38(3):276-284.
- Schwarz F, John G, Mainusch S, Sahm N, Becker J. Combined surgical therapy of peri-implantitis evaluating two methods of surface debridement and decontamination. A two-year clinical follow up report. J Clin Periodontol. 2012; 39(8):789-797.
- Sharma A. An ultraviolet-sterilization protocol for microtitre plates. J Exp Microbiol Immunol. 2012;16:144-147.
- World Medical Association Declaration of Helsinki: ethical principles for medical research involving human subjects. JAMA. 2013(20);310:2191-2194.
- Spencer P, Cobb CM, McCollum MH, Wieliczka DM. The effects of CO2 laser and Nd:YAG with and without water/ air surface cooling on tooth root structure: Correlation between FTIR spectroscopy and histology. J Periodontol Res. 1996;31(7):453-462.
- Jacques SL. Optical properties of biological tissues: a review. Phys Med Biol. 2013;58(11):R37-61.
- Zhang XQ, Bishop PL, Kupferle MJ. Measurement of polysaccharides and proteins in biofilm extracellular polymers. Water Sci Technol. 1998;37:345-348.
- Svaasand LO. Lasers for biomedical applications. In: Driggers RG, ed. Encyclopedia of Optical Engineering. New York, NY: Marcel Dekker; 2003:1035-1041.
- Vogel A, Venugopalan V. Mechanisms of pulsed laser ablation of biological tissues. Chem Rev. 2003;103(2):577-644.
- Einstein A. Über die von der molekularkinetischen Theorieder Wärme geforderte Bewegung von in ruhenden Flüssigkeiten suspendierten Teilchen. Annalen der Physik. 1905;322(8):549-60.
- Weast RC, ed. CRC Handbook of Chemistry and Physics. 61st ed. Boca Raton, FL: CRC Press; 1980-1981.
- Vitruk P. Oral soft tissue laser ablative and coagulative efficiencies spectra. Implant Practice US. 2014;7(6):22-27.
- Duplain G, Boulay R, Bélanger PA. Complex index of refraction of dental enamel at CO2 laser wavelengths. Appl Opt. 1987; 26(20):4447-4451.
- Wolfe WL, Zissis GJ. The Infrared Handbook. Washington DC: Office of Naval Research, NAVY, 1985:7-81.
Stay Relevant With Implant Practice US
Join our email list for CE courses and webinars, articles and mores





