Drs. Richard Horwitz, Eddie Scher, and Nikos Donos look at the appearance, prevention, and management of peri-implant diseases
Nowadays, peri-implant diseases are an increasing reality that we are confronted with in our profession. If they are not prevented, recognized early, or managed appropriately, they may lead to failure of the implant.
This article aims to provide an update into the nature of peri-implant disease, the diagnosis, and appropriate prevention and management.
Educational aims and objectives
This article aims to discuss the growing issue of peri-implantitis and present some suggestions for its prevention and management.
Expected outcomes
Implant Practice US subscribers can answer the CE questions to earn 2 hours of CE from reading this article and taking the quiz (click here for quiz). Correctly answering the questions will demonstrate the reader can:
• Identify the etiology of peri-implant diseases.
• Identify the steps needed to be taken during the clinical examination for diagnosis of
peri-implant disease.
• Recognize the risk factors and indicators of peri-implant disease.
• Realize the current thinking regarding management of peri-implant disease in patients.
• Realize the predictability of various treatment of peri-implant mucositis and peri-implantitis.
What are peri-implant diseases?
In general, peri-implant diseases are characterized by a host inflammatory response to opportunistic oral microbiota and are classified into peri-implant mucositis and peri-implantitis.
Peri-implant mucositis can be defined as a reversible inflammatory process residing in the soft tissues surrounding a functioning dental implant, whereas peri-implantitis is defined as an inflammatory process characterized by loss of surrounding bone in addition to the symptoms of the soft tissues (Lang, et al., 2011a).
How big a problem is this?
Dental implants present with very high survival rates (Albrektsson, et al., 1987; Pjetursson, et al., 2008). These, however, only report whether the implant is present at a certain moment in time, without evaluating whether it presents with biologic or technical complications.
At the same time, implant success includes various clinical and radiographic criteria (Ong, et al., 2008), which may often be reported using different terms (Needleman, et al., 2012).
The prevalence of peri-implantitis in the United Kingdom is still relatively unknown. Other reports have suggested that prevalence of peri-implantitis affects between 28% and 56% of severe chronic periodontitis patients and 12% and 43% of implant sites (Zitzmann and Berglundh, 2008). Another systematic review suggested prevalence at a patient level of 20% (Mombelli, et al., 2012).
How do we know if an implant is affected?
To diagnose peri-implant disease, the following steps need to take place during clinical examination:
Probing around the implant
The European Federation of Periodontology (EFP) consensus paper supports the use of a standard periodontal (UNC15) probe (Lang and Berglundh, 2011a). There is no conclusive evidence to demonstrate any detrimental effect that probing per se could pose around the implant (plastic or metal).
Probing needs to take place after loading of the implant, as a baseline, and at every maintenance visit.
If possible, removing the prosthesis can provide a more reliable probing depth reading (Serino and Ström, 2009).
Radiographical examination
Periapical radiographs should be taken at the time of loading the implant fixture so that a comparison can be made with future radiographs taken at assessment visits.
Following clinical change such as increased probing pocket depths, bleeding on probing or suppuration around an implant, an additional radiograph should also be taken, in order to evaluate the bone levels around this implant.
Bleeding on probing (BOP)
BOP has been shown to be a high negative predictor for implant disease (Jepsen, et al., 1996; Fransson, Wennström, and Berglundh, 2008). This means if there is no bleeding, there is a low probability of disease.
At the same time, suppuration is associated with active forms of disease, according to Fransson, Wennström, and Berglundh, especially in smokers (2008).
Peri-implantitis can be classified as an incidence of probing pocket depth (PPD) ≥5 mm with bleeding on probing and/or suppuration and radiographic signs of bone loss of ≥2.5mm or bone loss extending ≥ the first three threads (Ong, et al., 2008). However, other definitions exist in the literature.
Risk factors/indicators
Plaque
Plaque accumulation around dental implants was shown to increase the level of mucosal inflammation and pocket probing depths in an experimental study where patients were asked to refrain from cleaning for 3 weeks (Pontoriero, et al., 1994).
As such, it is now accepted that peri-implant mucositis is plaque-induced and that this may then progress to peri-implantitis if left untreated, as shown by Lang and colleagues (2011b).
Adequate oral hygiene measures and a cleansable prosthesis are very important in order to prevent peri-implant infection (Serino and Ström, 2009). Over-contoured restorations, poor pontic design, or close approximation of implants can provide obstacles for patients in the maintenance of their implants.
An observational study conducted in 2009 showed that approximately 50% of implants with peri-implantitis had a lack of accessibility for oral hygiene practices (Serino and Ström, 2009).
It has also been found that with poor oral hygiene, the patient presents with an odds ratio of approximately 14 times and is more likely to develop peri-implantitis (Ferreira, et al., 2006).
History of periodontal disease
There is a statistically significant risk of developing peri-implantitis in patients with periodontitis, even in those patients who are treated previously and declared as “perio-dontally stable.”
It has been calculated that in patients with previously treated periodontitis, the reported odds ratio of their developing peri-implantitis is 3.1 to 4.7 (Heitz-Mayfield and Huynh-Ba, 2009).
While this is significant, the reported implant survival rate for this cohort was still above 90%. Despite this, the success rates that indicate incidence of biological complications vary between studies from 58% to 100%. However, this is based on 5-year data, and different groups have suggested that the real long-term damage of peri-implantitis may present itself in survival data after 5 years (Heitz-Mayfield and Huynh-Ba, 2009).
Residual probing pocket depths ≥5 mm can serve as an even greater risk for peri-implant disease. Prevalence of peri-implantitis was found to be 11.8% to 13.3% in patients with a history of periodontitis (with no residual probing depths >5mm), compared to 30.1% of patients with active periodontitis (with periodontal probing depths >5mm) (Cho-Yan Lee, et al., 2012).
Smoking
Smoking not only has a negative impact on the periodontal tissues, but also has been shown to influence the prevalence and progression of peri-implant diseases (Roos-Jansåker, et al., 2006). Smokers have been shown to have a higher incidence of peri-implant bone loss in periodontal patients under maintenance (Fransson, Wennström, and Berglundh, 2008).
Diabetes
Patients with poorly controlled diabetes and periodontitis have been shown to present with an increase level of systemic “inflammation, oxidative stress, and apoptosis” (Retzepi, Lewis, and Donos, 2010; Genco and Borgnakke, 2013). This can contribute to destruction of periodontal tissues and impair wound healing, as shown by Genco and Borgnakke (2013).
Studies of patients with diabetes also reflect an increase in prevalence for peri-implantitis (Ferreira, et al., 2006).
The degree of glycemic control may also have a role, as early investigations seem to suggest that there may be limited clinical and microbiological differences in patients without diabetes and those with good glycemic control (Tatarakis, et al., 2014).
Other risk indicators, which we need to be aware of in our patients with implant fixtures, include but are not limited to genetic factors, alcohol consumption, systemic disease, and stress (Heitz-Mayfield, 2008; Dereka, et al., 2012).
How do we manage the patient with peri-implantitis?
Following diagnosis of peri-implant diseases, an appropriate management is vital in order to prevent deterioration of the peri-implant tissues. The goal of any treatment should be elimination/reduction of the pockets around the implants, with absence of suppuration, BOP, bone loss, and maintenance of healthy re-established peri-implant tissues (Heitz-Mayfield and Mombelli, 2014).
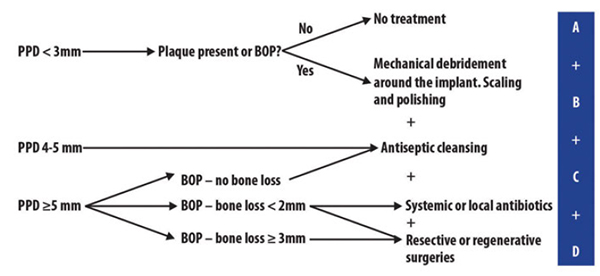
The Cumulative Interceptive Supportive Therapy (CIST) protocol has been suggested as a step-by-step guideline for the treatment of peri-implant diseases (Lang, et al., 2004).
The protocol, which is adapted in a flow diagram in Figure 1, provides guidance as to the stage/type of intervention that may be indicated at different clinical conditions. As probing depths increase along with BOP and bone loss, cumulative interventions are displayed in Figure 1 (A to D).
It also reinforces the need to take periapical radiographs when there are probing depths ≥5mm with bleeding on probing.
Other protocols have been written, including the Association of Dental Implantology (ADI) guidelines on treatment of peri-implant diseases (2012).
As peri-implant disease is bacterial in origin with contamination of the surface of the implant, mechanical debridement is the first line to any approach to therapy.
Many treatment modalities have been suggested for mechanical debridement and decontamination of the implant surfaces, including titanium curettes, plastic-coated ultrasonic inserts, air abrasion units, titanium brushes, and the use of chemical or photodynamic therapies — just some of the approaches that have been attempted.
How predictable is treatment?
Nonsurgical therapy has been shown to be effective in the treatment of peri-implant mucositis. However, treatment of peri-implantitis has shown a limited response (Klinge, et al., 2012).
The adjunctive uses of local or systemic antibiotics have been shown to reduce bleeding on probing and pocket depth, but with modest outcomes (Renvert, et al., 2008).
Due to the different implant surface characteristics and therefore limited access to the biofilm, surgical treatment may be indicated at an earlier stage and more frequently compared to conventional periodontal therapy (Heitz-Mayfield and Lang, 2010).
et al., 2012)
Summary
Preventive approach should start in the implant planning stages and case selection.
It is important to assess the patient risk profile so that maintenance can be targeted appropriately.
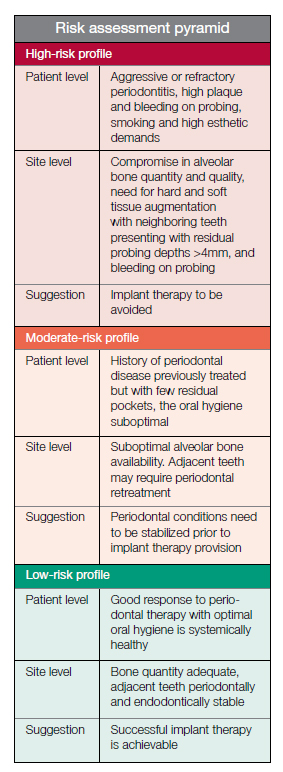
Figure 2 (Donos, Laurell, and Mardas, 2012) demonstrates these risk profiles in a simple way for us to start this preventive process and case selection from the early stages in treatment planning. The majority of patients would fit into this lower risk category.
It is of upmost importance to continually monitor the health of our patients and their implants. Regular maintenance visits are essential depending on the risk profile of the patient. Supportive periodontal therapy is vital in the prevention of peri-implant disease, as reported by Donos, Laurell, and Mardas (2012).
A summary of what can be included in the visit is shown as follows:
- Parallel periapical radiographs taken immediately post-implant fixture placement at the time of loading and at the 1-year review appointment (Lang and Berglundh, 2011a)
- Detailed oral hygiene advice, demonstrating how to perform efficient plaque removal
- Depending on the severity of perio-dontal disease and presence of risk factors for disease progression, examination should take place every 3, 6, or 12 months. This should include PPD, BOP, and plaque scores (Donos, Laurell, and Mardas, 2012)
- Further radiographic examination should take place in the presence of a peri-implant site with BOP and PPD ≥5 mm (Lang, et al., 2004)
- Professional hygiene treatments including supra- and subgingival debridement should be performed every 3 to 6 months with appropriate instruments (Donos, Laurell, and Mardas, 2012)
Following a management protocol is vital in preventing peri-implant diseases. This empowers the clinician to recognize this condition and manage appropriately.
References
- Albrektsson T, Dahl E, Enbom L, Engevall S, Engquist B, Eriksson AR, Feldmann G, Freiberg N, Glantz PO, Kjellman O et al. Osseointegrated oral implants. A Swedish multicenter study of 8139 consecutively inserted Nobelpharma implants. J Periodontol. 1988;59(5):287-296.
- Cho-Yan Lee J, Mattheos N, Nixon KC, Ivanovski S. Residual periodontal pockets are a risk indicator for peri-implantitis in patients treated for periodontitis. Clin Oral Implants Res. 2012;23(3):325–333.
- Dereka X, Mardas N, Chin S, Petrie A, Donos N. A systematic review on the association between genetic predisposition and dental implant biological complications. Clin Oral Implants Res. 2012;23(7):775–788.
- Donos N, Laurell L, Mardas N. Hierarchical decisions on teeth vs. implants in the periodontitis-susceptible patient: the modern dilemma. Periodontol 2000. 2012;59(1):89–110.
- Ferreira SD, Silva GL, Cortelli JR, Costa JE, Costa FO. Prevalence and risk variables for peri-implant disease in Brazilian subjects. J Clin Periodontol. 2006;33(12):929–935.
- Fransson C, Wennström J, Berglundh T. Clinical characteristics at implants with a history of progressive bone loss. Clin Oral Implants Res. 2008;19(2):142–147.
- Genco RJ, Borgnakke WS. Risk factors for periodontal disease. Periodontol 2000. 2013;62(1):59–94.
- Heitz-Mayfield LJ, Lang NP. Comparative biology of chronic and aggressive periodontitis vs peri-implantitis. Periodontol 2000. 2010;53:167–181.
- Heitz-Mayfield LJ, Mombelli A. The therapy of peri-implantitis: a systematic review. Int J Oral Maxillofac Implants. 2014;(29 Suppl):325–345.
- Heitz-Mayfield LJ. Peri-implant diseases: diagnosis and risk indicators. J Clin Periodontol. 2008;35(8 Suppl):292–304.
- Heitz-Mayfield LK, Huynh-Ba G. History of treated periodontitis and smoking as risks for implant therapy. Int J Oral Maxillofac Implants. 2009;(24 Suppl):39–68.
- Klinge B, Meyle J, Working Group 2. Peri-implant tissue destruction. The Third EAO Consensus Conference 2012. Clin Oral Implants Res. 2012;23(6 Suppl):108–110.
- Lang NP, Berglundh T, Heitz-Mayfield LJ, Pjetursson BE, Salvi GE, Sanz M. Consensus statements and recommended clinical procedures regarding implant survival and complications. Int J Oral Maxillofac Implants. 2004;19 Suppl:150–154.
- Lang NP, Berglundh T, Working Group 4 of Seventh European Workshop on Periodontology. Periimplant diseases: Where are we now? – Consensus of the Seventh European Workshop on Periodontology. J Clin Periodontol. 2011;38(11 Suppl):178–181.
- Lang NP, Bosshardt DD, Lulic M. Do mucositis lesions around implants differ from gingivitis lesions around teeth? J Clin Periodontol. 2011;38 (11 Suppl):182–187.
- Mombelli A, Müller N, Cionca N. The epidemiology of peri-implantitis. Clin Oral Implants Res. 2012;23(6 Suppl):67-76.
- Needleman I, Chin S, O’Brien T, Petrie A, Donos N. Systematic review of outcome measurements and reference group(s) to evaluate and compare implant success and failure. J Clin Periodontol. 2012;39(12 Suppl):122–132.
- Ong CT, Ivanovski S, Needleman IG, Retzepi M, Moles DR, Tonetti MS, Donos N. Systematic review of implant outcomes in treated periodontitis subjects. J Clin Periodontol. 2008;35(5):438–462.
- Pjetursson BE, Tan WC, Zwahlen M, Lang NP. A systematic review of the success of sinus floor elevation and survival of implants inserted in combination with sinus floor elevation. J Clin Periodontol. 2008;35(8 Suppl):216–240.
- Pontoriero R, Tonelli MP, Carnevale G, Mombelli A, Nyman SR, Lang NP. Experimentally induced peri-implant mucositis. A clinical study in humans. Clin Oral Implants Res. 1994;5(4):254–259.
- Renvert S, Roos-Jansåker AM, Claffey N. Non-surgical treatment of peri-implant mucositis and peri-implantitis: A literature review. J Clin Periodontol. 2008;35(8 Suppl):305–315.
- Retzepi M, Lewis MP, Donos N. Effect of diabetes and metabolic control on de novo bone formation following guided bone regeneration. Clin Oral Implants Res. 2010;21(1):71–79.
- Roos-Jansåker AM, Renvert H, Lindahl C, Renvert S. Nine- to fourteen-year follow-up of implant treatment. Part III: Factors associated with peri-implant lesions. J Clin Periodontol. 2006;33(4):296–301.
- Jepsen S, Rühling A, Jepson K, Ohlenbusch B, Albers HK. Progressive peri-implantitis. Incidence and prediction of peri-implant attachment loss. Clin Oral Implants Res. 1996;7(2):133–142.
- Serino G, Ström C. Peri-implantitis in partially edentulous patients: association with inadequate plaque control. Clin Oral Implants Res. 2009;20(2):169–174.
- Tatarakis N, Kinney JS, Inglehart M, Braun TM, Shelburne C, Lang NP, Giannobile WV, Oh TJ. Clinical, microbiological, and salivary biomarker profiles of dental implant patients with type 2 diabetes. Clin Oral Implants Res. 2014;25(7):803–812.
- Zitzmann NU, Berglundh T. Definition and prevalence of peri-implant diseases. J Clin Periodontol. 2008;35(8 Suppl):286-91.
Stay Relevant With Implant Practice US
Join our email list for CE courses and webinars, articles and mores


