In the first of two articles, Dr. Paul Tipton introduces some of the advances in restorative materials with a discussion of polyetheretherketone — a new prosthodontic material
The high-performance polymers (HPPs) are the uppermost class of plastics, possessing better temperature and chemical stability and mechanical properties than the commodity plastics, but typically
being manufactured in lower volumes and costing more.
Educational aims and objectives
The aim of this article is to explain the material properties and clinical potential of the high-performance polymer family of plastics within implant dentistry.
Expected outcomes
Implant Practice US subscribers can answer the CE questions to earn 2 hours of CE from reading this article. Correctly answering the questions will demonstrate the reader can:
- Identify the scientific principles underlying the PAEK family of materials.
- Realize some uses of PAEKs in the medical practice
- Realize some uses of PAEKs in the dental practice.
- Identify types of PAEKs for prosthetics.
- Realize some methods of framework manufacture.

 The family of HPPs that have entered dentistry are called the polyarletherketones (PAEKs), of which there are several members with varying chemical structures. Many of us in the dental industry are inadvertently familiar with the family member called PEEK (polyetheretherketone) through its use in healing caps, temporary abutments, and scan bodies.
The family of HPPs that have entered dentistry are called the polyarletherketones (PAEKs), of which there are several members with varying chemical structures. Many of us in the dental industry are inadvertently familiar with the family member called PEEK (polyetheretherketone) through its use in healing caps, temporary abutments, and scan bodies.
However, the reason for the recent enthusiasm surrounding PAEKs has been their potential for use as a metal alternative in broader indications such as removable dentures (Figures 1 and 2) and implant-borne prosthetics (Figure 3). It is here that the shock-absorbing characteristics of the material could be extremely interesting for immediate loading or long-term frameworks (Figure 4).
This article will describe the background to these materials.
The PAEK family
PEEK is the most well-known and most widely used member of the PAEK family. PEEK was invented in the United Kingdom in 1978 (ICI – now trading as Victrex) and was selected by aerospace, semiconductor, automotive, and medical industries as a standard material of use in all these sectors.
It is typically used as a metal replacement, due to its strength-to-weight ratio and corrosion resistance. Other family members also exist, which are variations of the chemistry (e.g., PEK and PEKK), and these materials can also be filled with pigments or reinforcing agents. In their unaltered, unfilled state, the materials are beige in color.
PAEKs in medical
Several of the properties of PEEK that were being exploited in industry (such as their strength-to-weight ratio, chemical and wear resistance, radiolucency, and reduced stiffness versus metals) were naturally intriguing for medical use. The first published PEEK medical research came in the 1980s (Williams, et al., 1987) followed by the first implantable grade from Invibio™ Biomaterial Solutions in the 1990s (Victrex/Invibio, United Kingdom).
Medical grades have a much tighter specification and increased quality control than industrial grade materials, which is important in the wake of the silicone breast implant scandal. (See https://www.nhs.uk/conditions/breast-implants/pages/pip-introduction.aspx.)
PEEK remained the only medical PAEK for many years. Spine surgeons in particular adopted Invibio’s PEEK, liking the reduced Young’s Modulus (stiffness) of the material and the scatter-free CT and MRI imaging. PEEK has since become the standard alternative to titanium for load-bearing spinal cage devices for the spine. Today, manufacturer Invibio claims PEEK has been used in around 5 million implantable devices, spanning some 500 separate US FDA 510(k) regulatory clearances. In more recent years, additional versions of PEEK and PEKK have appeared on the medical marketplace but have been limited in use.
PAEKs in dentistry
Short-term devices such as temporary healing caps and abutments have been sold directly to dentists through the dental companies for many years. In these situations, either unfilled PEEK or PEEK with a 10% titanium dioxide pigment filler is typical and has been used in these temporary devices for over a decade.
In the case of customized prostheses, the upstream material or shape becomes the “device” and is regulated and cleared for use for a defined set of indications. Here, the PAEKs have appeared as materials for use in injection press systems or as discs for computer-aided design/manufacture (CAD/CAM).
Types of PAEKs for prosthetics
There are now many brands of PAEK dental devices becoming available for use in prosthetic frameworks. The most common formulations of the PAEKs are:
- Unfilled, pure 100% PEEK (such as JUVORA™ [Invibio/Juvora]). This is a beige material.
- 80% PEEK with 20% nanoceramic filler (such as BioHPP [Bredent]). This is a white material.
- 80% PEEK with 20% titanium-dioxide filler (such as Dentokeep disc [NT Trading]). This is a white material.
- 80% PEKK with 20% filler including titanium dioxide (such as Pekkton® ivory [Cendres & Métaux]). This is an off-white material.
Typically, the particle size of these fillers (circa 300-500 nanometers for the nano-ceramic) is not likely to give significant reinforcing properties to the material, since they are not fibers. Instead the fillers act more as a pigment and alter surface topography. These levels of 20% filler will make the material stiffness slightly higher, but consequently also slightly increase its brittleness.
It should also be noted that the inclusion of titanium dioxide means that these brands — Biohpp, Dentokeep, and Pekkton — should not be pitched as “metal free” since this could be in breach of Advertising Standards and/or governing bodies.
The reader should also take note as to the cleared indications for use, as the different materials and forms may have varying clearances. To date, PAEKs with these specific 20% fillers have only a limited history of use in dental and actually no prior medical history in any other medical applications. Therefore, it is fair to say that the jury is still out as to the effects of adding these levels of these specific fillers to the PAEKs, and the author advises the use only of the pure material where there are long-term studies.
Methods of framework manufacture
There are two methods for laboratories to manufacture substructure frameworks from PAEKs. These are 1) injection molding, or 2) CAD/CAM.
Injection molding
Industrial injection molding machines process the polymer under very high speed and pressure (with a bar pressure in the thousands), which are typically two orders of magnitude higher than the typical benchtop pressing machines available to the dental laboratory (which have a bar pressure in the 10s).
This means that small-scale injection molding of PAEK is no mean feat, due to tight processing windows and design limitations. Also, these re-melting of PAEKs can also increase the risk of unpredictable mechanical and physical properties (such as brittleness, flexibility, color, warping) if the framework has not cooled and recrystallized correctly. Finally, re-melting of PAEK materials can also cause degradation of the polymer (such as generation of phenol) unless very closely controlled using the correct equipment.
This polymer degradation can be accentuated by the inclusion of fillers in the materials (such as reinforcing agents or pigments). Therefore, melt processing of these materials should only be done by a competent laboratory and using the equipment recommended by the supplier.
CAD/CAM
The alternative manufacture route uses CAD/CAM technology. This manufacturing route avoids all of the risks mentioned previously for re-melting the polymer. The material properties remain consistent, and the framework manufacture can also benefit from the increase precision and reproducibility of a digital workflow.
Although it does require a more significant capital investment by the laboratory, many laboratories are seeing that it is necessary to align with other industries and adopt digitization to increase efficiencies. PAEK materials further extend these CAD/CAM efficiencies when compared to milling metal substructures, since there is typically less tool wear and faster milling times, and the capital equipment necessary to mill them does not need to be as expensive as machines for milling metal frameworks. It is the author’s view that the optimum use of these materials only comes from the CAD/CAM milling process as opposed to the injection
molding process.
Polymer properties
When handling a prosthetic framework made from a PAEK, a striking thing is the difference in weight. When identical full-arch implant prosthetic substructure frameworks were made from four different materials, the results from weighing were:
- PAEK — 4.9 g
- Titanium — 17 g
- Zirconia — 23 g
- Cobalt chrome — 33 g
However, it is the possibility to introduce shock absorption to a prosthesis that is perhaps the most exciting. This could have positive implications for patient comfort and for damage limitation.
In the author’s view, the most relevant mechanical property related to the aspect of shock absorption is not ultimately compressive strength (as is sometimes promoted), but actually flexural strength and elastic modulus. Obtaining an increasingly stronger material becomes academic since clearly it would be simplistic to prefer the highest value.
Metals have very high compressive strengths relative to PAEKs but are not shock absorbing. Naturally, a design must also consider the influence of thickness and shape as well, but values for flexural strength and elastic modulus are more indicative of the stiffness of a material and how much it will deflect the load.
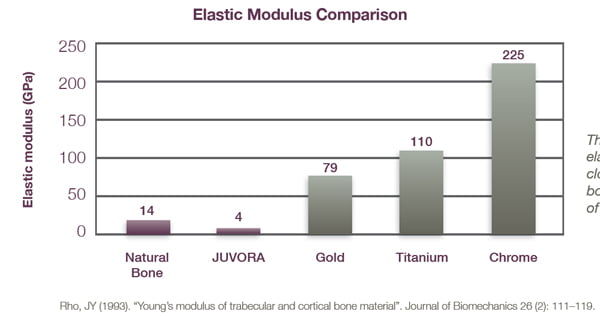
Stiffer materials, like metals, have a high elastic modulus (see Figure 5) meaning that metals require high loads to elastically deform them. Therefore, one can look at natural materials like bone for clues as to an ideal for stiffness.
Common denture materials like PMMA have an elastic modulus range of 1.8-3.1 GPa but limited strength. The PAEKs have an elastic modulus closer to bone (4-5GPa) allowing the framework to be stiffer, yet still shock absorbing. However, PAEKs also additionally possess sufficient strength to be considered as a metal alternative.
 Conclusions
Conclusions
The high-performance polymers called PEEK and PEKK have exciting potential in dentistry as a metal alternative for removable and implant prosthetic frameworks. Their stiffness properties confer promise as a substructure that could add an element of shock absorption. This may have benefits for patient comfort, addressing parafunction, and damage limitation.
References
Williams DF, McNamara A, Tutner RM. Potential of poly ether ether ketone (PEEK) and carbon-fiber-reinforced PEEK in medical applications. Journal of Materials Science Letters. 1987;6(2):188-190.



